Nitrene
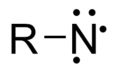
In chemistry, a nitrene (R–N:) is the nitrogen analogue of a carbene. The nitrogen atom has only 5 valence electrons and is therefore considered an electrophile. A nitrene is a reactive intermediate and is involved in many chemical reactions.[1][2]
Electron configuration
In the most simple nitrene, the linear imidogen (:N–H), two of the 6 available electrons form a covalent bond with hydrogen, two other create a free electron pair and the two remaining electrons occupy two degenerate p orbitals. Consistent with Hund's rule the low energy form of imidogen is a triplet with one electron in each of the p orbitals and the high energy form is the singlet state with an electron pair filling one p orbital and the other one vacant.
As with carbenes, a strong correlation exists between the spin density on the nitrogen atom which can be calculated in silico and the zero-field splitting parameter D which can be derived experimentally from electron spin resonance.[3] Small nitrenes such as NH or CF3N have D values around 1.8 cm−1 with spin densities close to a maximum value of 2. At the lower end of the scale are molecules with low D (< 0.4) values and spin density of 1.2 to 1.4 such as 9-anthrylnitrene and 9-phenanthrylnitrene.
Formation
Because nitrenes are so reactive, they are not isolated. Instead, they are formed as reactive intermediates during a reaction. There are two common ways to generate nitrenes:
- From azides by thermolysis or photolysis, with expulsion of nitrogen gas. This method is analogous to the formation of carbenes from diazo compounds.
- From isocyanates, with expulsion of carbon monoxide. This method is analogous to the formation of carbenes from ketenes.
Reactions
Nitrene reactions include:
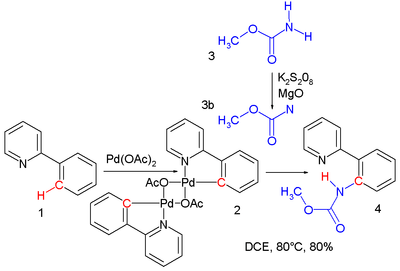
- Nitrene C–H insertion. A nitrene can easily insert into a carbon to hydrogen covalent bond yielding an amine or amide. A singlet nitrene reacts with retention of configuration. In one study[4] a nitrene, formed by oxidation of a carbamate with potassium persulfate, gives an insertion reaction into the palladium to nitrogen bond of the reaction product of palladium(II) acetate with 2-phenylpyridine to methyl N-(2-pyridylphenyl)carbamate in a cascade reaction:
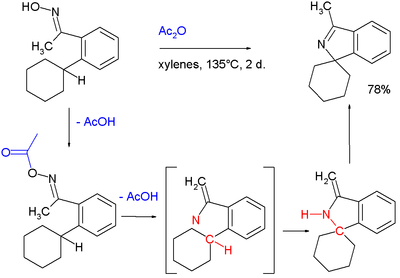
- A nitrene intermediate is suspected in this C–H insertion involving an oxime, acetic anhydride leading to an isoindole:[5]
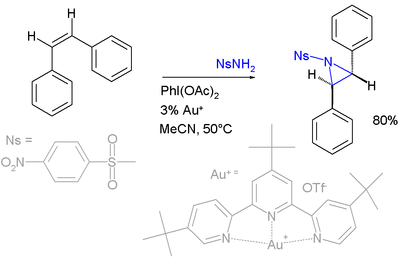
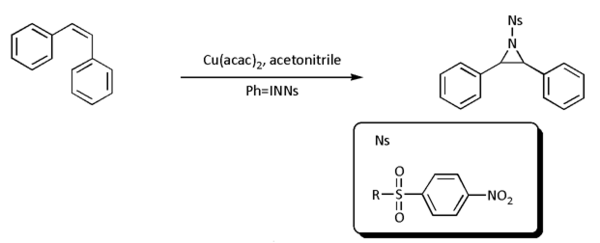
- Nitrene cycloaddition. With alkenes, nitrenes react to aziridines, very often with nitrenoid precursors such as nosyl- or tosyl-substituted [N-(phenylsulfonyl)imino]phenyliodinane (PhI=NNs or PhI=NTs respectively)) but the reaction is known to work directly with the sulfonamide in presence of a transition metal based catalyst such as copper, palladium, or gold:[6][7][8][9][10]
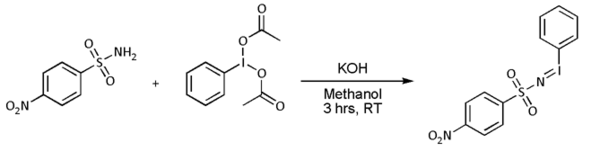
- In most cases, however, [N-(p-nitrophenylsulfonyl)imino]phenyliodinane (PhI=NNs) is prepared separately as follows:
- Nitrene transfer takes place next:
- In this particular reaction both the cis-stilbene illustrated and the trans form (not depicted) result in the same trans-aziridine product, suggesting a two-step reaction mechanism. The energy difference between triplet and singlet nitrenes can be very small in some cases, allowing interconversion at room temperature. Triplet nitrenes are thermodynamically more stable but react stepwise allowing free rotation and thus producing a mixture of stereochemistry.[11]
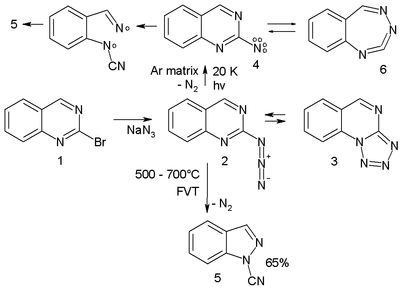
- Arylnitrene ring-expansion and ring-contraction. Aryl nitrenes show ring expansion to 7-membered ring cumulenes, ring opening reactions and nitrile formations many times in complex reaction paths. For instance the azide 2 in the scheme below[3] trapped in an argon matrix at 20 K on photolysis expels nitrogen to the triplet nitrene 4 (observed experimentally with ESR and ultraviolet-visible spectroscopy) which is in equilibrium with the ring-expansion product 6.
- The nitrene ultimately converts to the ring-opened nitrile 5 through the diradical intermediate 7. In a high-temperature reaction, FVT at 500–600 °C also yields the nitrile 5 in 65% yield.[12]
Nitreno radicals
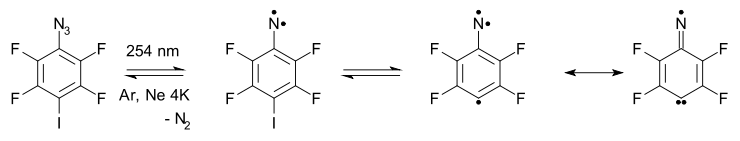
For several compounds containing both a nitrene group and a free radical group an ESR high-spin quartet has been recorded (matrix, crygenic temperatures). One of these has an amine oxide radical group incorporated,[13] another system has a carbon radical group.[14]
In this system one of the nitrogen unpaired electrons is delocalized in the aromatic ring making the compound a σ–σ–π triradical. A carbene nitrogen radical (imidyl radical) resonance structure makes a contribution to the total electronic picture.
References
- ↑ Lwowski, W., ed. (1970). Nitrenes. New York: Interscience.
- ↑ Wentrup, C. (1984). Reactive Intermediates. New York: Wiley.
- 1 2 Kvaskoff, David; Bednarek, Paweł; George, Lisa; Waich, Kerstin; Wentrup, Curt (2006). "Nitrenes, Diradicals, and Ylides. Ring Expansion and Ring Opening in 2-Quinazolylnitrenes". J. Org. Chem. 71 (11): 4049–4058. doi:10.1021/jo052541i.
- ↑ Thu, Hung-Yat; Yu, Wing-Yiu; Che, Chi-Ming (2006). "Intermolecular Amidation of Unactivated sp2 and sp3 C–H Bonds via Palladium-Catalyzed Cascade C–H Activation/Nitrene Insertion". J. Am. Chem. Soc. 128 (28): 9048–9049. doi:10.1021/ja062856v.
- ↑ Savarin, Cécile G.; Grisé, Christiane; Murry, Jerry A.; Reamer, Robert A.; Hughes, David L. (2007). "Novel Intramolecular Reactivity of Oximes: Synthesis of Cyclic and Spiro-Fused Imines". Org. Lett. 9 (6): 981–983. doi:10.1021/ol0630043.
- ↑ Li, Zigang; Ding, Xiangyu; He, Chuan (2006). "Nitrene Transfer Reactions Catalyzed by Gold Complexes". J. Org. Chem. 71 (16): 5876–5880. doi:10.1021/jo060016t.
- ↑ Evans, David A.; Faul, Margaret M.; Bilodeau, Mark T. (1994). "Development of the Copper-Catalyzed Olefin Aziridination Reaction". J. Am. Chem. Soc. 116: 2742–2753. doi:10.1021/ja00086a007.
- ↑ Brandt, Peter; Sodergren, Mikael J.; Andersson, Pher G.; Norrby, Per-Ola (2000). "Mechanistic Studies of Copper-Catalyzed Alkene Aziridination". J. Am. Chem. Soc. 112: 8013–8020.
- ↑ Watson, Iain D. G.; Yu, Lily; Yudi, Andrei K. (2006). "Advances in Nitrogen Transfer Reactions Involving Aziridines". Acc. Chem. Res. 39: 194–206. doi:10.1021/ar050038m.
- ↑ Reactants cis-stilbene or trans-stilbene, nitrene precursor p-nitrosulfonamide or nosylamine which is oxidized by iodosobenzene diacetate. The gold catalyst is based on a terpyridine tridentate ligand.
- ↑ Yudin, Andrei K., ed. (2007). Aziridines and Epoxides in Organic Synthesis. p. 120. ISBN 3-527-31213-7.
- ↑ The quinazoline is prepared from the corresponding bromide and sodium azide. The azide is in equilibrium with the tetrazole 3.
- ↑ Lahti, Paul M.; Esat, Burak; Liao, Yi; Serwinski, Paul; Lan, Jiang; Walton, Richard (30 May 2001). "Heterospin organic molecules: nitrene–radical linkages". Polyhedron. 20 (11–14): 1647–1652. doi:10.1016/S0277-5387(01)00667-2.
- ↑ Sander, Wolfram; Grote, Dirk; Kossmann, Simone; Neese, Frank (2008). "2,3,5,6-Tetrafluorophenylnitren-4-yl: Electron Paramagnetic Resonance Spectroscopic Characterization of a Quartet-Ground-State Nitreno Radical". J. Am. Chem. Soc. 130: 4396–4403. doi:10.1021/ja078171s.