Catellani reaction
The Catellani reaction was discovered by Marta Catellani (Università degli Studi di Parma, Italy) and co-workers in 1997.[1][2] The reaction uses aryl iodides to perform bi- or tri-functionalization, including C-H functionalization of the unsubstituted ortho position(s), followed a terminating cross-coupling reaction at the ipso position. This cross-coupling cascade reaction depends on the ortho-directing transient mediator, norbornene.
Reaction mechanism
The Catellani reaction is catalyzed by palladium and norbornene, although in most cases superstochiometric amounts of norbornene to deduce reaction time.[3] Palladium(0) oxidatively inserts in to aryl-iodide bond. Once carbopallation with the norbornene, the structure of the norbornylpalladium intermediate does not allow for ß-hydride elimination in either of the ß-positions due to Bredt's Rule over bridgehead proton and the trans-configuration of the palladium and ß-proton.[4] Thereafter, the Pd(II) species undergoes electrophilic cyclopalladation at the ortho position. The second oxidative addition occurs with the corresponding coupling partner to form a Pd(IV) intermediate, which then reductively eliminates and completes the first cross-coupling bond formation. After the elimination of norbornene, the Pd(II) species undergoes the second cross-coupling reaction, terminating the series with a number of traditional palladium-catalyzed coupling reactions.[5]
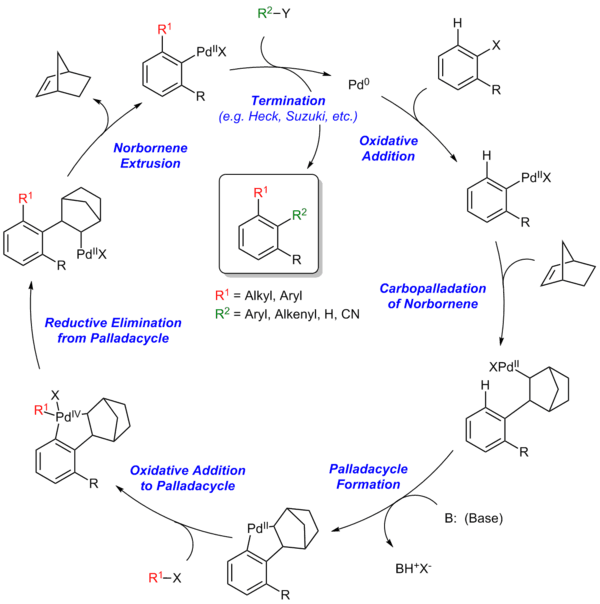
Steps of the Catellani reaction:
- Oxidative addition
- Carbopalladation of norbornene
- Palladacycle formation
- Oxidative addition to palladacycle
- Reductive elimination from palladacycle
- Norbornene extrusion
- Termination via Heck reaction, Suzuki reaction, etc.
Ortho and ipso cross-coupling partners
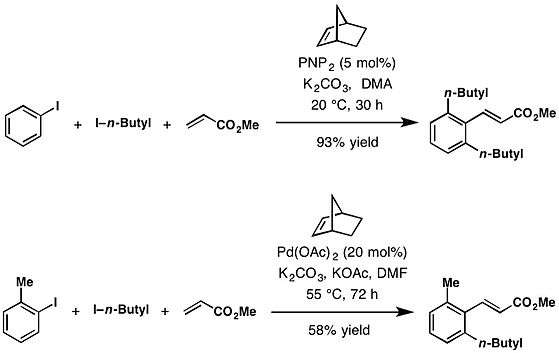
The Catellani reaction facilitates a variety of C—C and C—N bond-forming reactions at the ortho position. These include alkylation from alkyl halides,[1] arylation from aryl bromides,[7] amination from benzyloxyamines,[8][9][10][11] acylation from anhydrides.[12][13] Likewise in the case of terminating ipso coupling partners with Heck-type termination with olefins,[1] Suzuki-type reaction with boronic esters,[8] borylation with bis(pinacolato)diboron,[9] protonation with i-PrOH,[10] decarboxylative alkynylation with alkynyl carboxylic acids.[11]
Uses
With tethered cross-coupling partners, Lautens, Malacria, and Catellani used this reaction to construct a variety of fused ring systems since 2000.[5] The Catellani reaction has been used as a key step for the total synthesis (+)-linoxepin,[14] rhazinal,[15] aspidospermidine,[16] and (±)-goniomitine.[16]
References
- 1 2 3 Catellani, Marta; Frignani, Franco; Rangoni, Armando (1997-02-03). "A Complex Catalytic Cycle Leading to a Regioselective Synthesis of o,o′-Disubstituted Vinylarenes". Angewandte Chemie International Edition in English. 36 (1-2): 119–122. doi:10.1002/anie.199701191. ISSN 1521-3773.
- ↑ Catellani; et al. (1997). "Regioselektive Synthese o,o′-disubstituierter Vinylarene über einen komplexen Katalysecyclus". Angewandte Chemie. 109 (1-2). doi:10.1002/ange.19971090146. Retrieved 26 December 2014.
- ↑ Catellani, Marta; Motti, Elena; Della Ca’, Nicola (2008-11-18). "Catalytic Sequential Reactions Involving Palladacycle-Directed Aryl Coupling Steps". Accounts of Chemical Research. 41 (11): 1512–1522. doi:10.1021/ar800040u. ISSN 0001-4842.
- ↑ Martins; et al. (2010). "Synthesis in the Key of Catellani: Norbornene-Mediated ortho C–H Functionalization". Top Curr Chem. 292. doi:10.1007/128_2009_13. Retrieved 25 November 2015.
- 1 2 Ye, Juntao; Lautens, Mark. "Palladium-catalysed norbornene-mediated C–H functionalization of arenes". Nature Chemistry. 7 (11): 863–870. doi:10.1038/nchem.2372.
- ↑ Martins; et al. (2010). "Synthesis in the Key of Catellani: Norbornene-Mediated ortho C–H Functionalization". Top Curr Chem. 292. doi:10.1007/128_2009_13. Retrieved 25 November 2015.
- ↑ Faccini, Fiorenza; Motti, Elena; Catellani, Marta (2004-01-01). "A New Reaction Sequence Involving Palladium-Catalyzed Unsymmetrical Aryl Coupling". Journal of the American Chemical Society. 126 (1): 78–79. doi:10.1021/ja039043g. ISSN 0002-7863.
- 1 2 Ye, Changqing; Zhu, Hui; Chen, Zhiyuan (2014-09-19). "Synthesis of Biaryl Tertiary Amines through Pd/Norbornene Joint Catalysis in a Remote C–H Amination/Suzuki Coupling Reaction". The Journal of Organic Chemistry. 79 (18): 8900–8905. doi:10.1021/jo501544h. ISSN 0022-3263.
- 1 2 Shi, Hang; Babinski, David J.; Ritter, Tobias (2015-03-25). "Modular C–H Functionalization Cascade of Aryl Iodides". Journal of the American Chemical Society. 137 (11): 3775–3778. doi:10.1021/jacs.5b01082. ISSN 0002-7863.
- 1 2 Dong, Zhe; Dong, Guangbin (2013-12-11). "Ortho vs Ipso: Site-Selective Pd and Norbornene-Catalyzed Arene C–H Amination Using Aryl Halides". Journal of the American Chemical Society. 135 (49): 18350–18353. doi:10.1021/ja410823e. ISSN 0002-7863.
- 1 2 Sun, Fenggang; Gu, Zhenhua (2015-05-01). "Decarboxylative Alkynyl Termination of Palladium-Catalyzed Catellani Reaction: A Facile Synthesis of α-Alkynyl Anilines via Ortho C–H Amination and Alkynylation". Organic Letters. 17 (9): 2222–2225. doi:10.1021/acs.orglett.5b00830. ISSN 1523-7060.
- ↑ Huang, Yunze; Zhu, Rui; Zhao, Kun; Gu, Zhenhua (2015-10-19). "Palladium-Catalyzed Catellani ortho-Acylation Reaction: An Efficient and Regiospecific Synthesis of Diaryl Ketones". Angewandte Chemie International Edition. 54 (43): 12669–12672. doi:10.1002/anie.201506446. ISSN 1521-3773.
- ↑ Zhou, Ping-Xin; Ye, Yu-Ying; Liu, Ce; Zhao, Lian-Biao; Hou, Jian-Ye; Chen, Dao-Qian; Tang, Qian; Wang, An-Qi; Zhang, Jie-Yu (2015-08-07). "Palladium-Catalyzed Acylation/Alkenylation of Aryl Iodide: A Domino Approach Based on the Catellani–Lautens Reaction". ACS Catalysis. 5 (8): 4927–4931. doi:10.1021/acscatal.5b00516.
- ↑ Weinstabl; et al. (16 Apr 2013). "Total Synthesis of (+)-Linoxepin by Utilizing the Catellani Reaction". Angewandte Chemie International Edition. 52 (20). doi:10.1002/anie.201302327. Retrieved 26 December 2014.
- ↑ Sui; et al. (June 2013). "Pd-Catalyzed Chemoselective Catellani Ortho-Arylation of Iodopyrroles: Rapid Total Synthesis of Rhazinal". J. Am. Chem. Soc. 135 (25): 9318–9321. doi:10.1021/ja404494u. Retrieved 26 December 2014.
- 1 2 Jiao, Lei; Herdtweck, Eberhardt; Bach, Thorsten (2012-09-05). "Pd(II)-Catalyzed Regioselective 2-Alkylation of Indoles via a Norbornene-Mediated C–H Activation: Mechanism and Applications". Journal of the American Chemical Society. 134 (35): 14563–14572. doi:10.1021/ja3058138. ISSN 0002-7863.