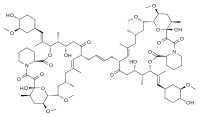
FK1012
 | |
| Names | |
|---|---|
| IUPAC name
(1R,12S,13R,14S,18E,21S,23S,24R,25R,27R)-1,14-Dihydroxy-17-{(2E)-4-[(12S,13R,14S,17R,18E,21S,23S,24R,25R,27R)-14-hydroxy-12-{(1E)-1-[(1R,3R,4R)-4-hydroxy-3-methoxycyclohexyl]-1-propen-2-yl}-23,25-dimethoxy-13,19,21,27-tetramethyl-2,3,10,16-tetraoxo-11,28-dioxa-4-azatricyclo[22.3.1.04,9]octacos-18-en-17-yl]-2-buten-1-yl}-12-{(1E)-1-[(1R,3R,4R)-4-hydroxy-3-methoxycyclohexyl]-1-propen-2-yl}-23,25-dimethoxy-13,19,21,27-tetramethyl-11,28-dioxa-4-azatricyclo[22.3.1.04,9]octacos-18-ene-2,3,10,16-tetrone | |
| Identifiers | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 23193198 |
| |
| |
| Properties | |
| C86H134N2O23 | |
| Molar mass | 1,564.01 g·mol−1 |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
FK1012, a derivative of tacrolimus, is used as a research tool in chemically induced dimerization applications. The protein FKBP does not normally form dimers but can be caused to dimerize in the presence of FK1012. Genetically engineered proteins based on FKBP can be used to manipulate protein localization, signalling pathways and protein activation.[1]
FK1012 is a dimer of tacrolimus; the two tacrolimus units are linked at their vinyl groups.[2]
References
- ↑ Fegan, A; White, B; Carlson, JC; Wagner, CR (Jun 9, 2010). "Chemically controlled protein assembly: techniques and applications.". Chemical Reviews. 110 (6): 3315–36. doi:10.1021/cr8002888. PMID 20353181.
- ↑ Steven T. Diver, Stuart L. Schreiber (1997). "Single-step syntheses of cell permeable protein dimerizers that activate signal transduction and gene expression". J. Am. Chem. Soc. 119: 5106–5109.
Further reading
- Otto, K. G.; Jin, L; Spencer, D. M.; Blau, C. A. (2001). "Cell proliferation through forced engagement of c-Kit and Flt-3". Blood. 97 (11): 3662–4. doi:10.1182/blood.V97.11.3662. PMID 11369667.
This article is issued from Wikipedia - version of the 11/9/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.