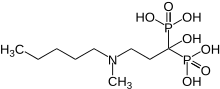
Ibandronic acid
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Oral, intravenous |
| ATC code | M05BA06 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 0.6% |
| Protein binding |
90.9 to 99.5% (concentration-dependent) |
| Metabolism | Nil |
| Biological half-life | 10 to 60 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number |
114084-78-5 |
| PubChem (CID) | 60852 |
| IUPHAR/BPS | 3059 |
| DrugBank |
DB00710 |
| ChemSpider |
54839 |
| UNII |
UMD7G2653W |
| KEGG |
D08056 |
| ChEMBL |
CHEMBL997 |
| ECHA InfoCard | 100.214.537 |
| Chemical and physical data | |
| Formula | C9H23NO7P2 |
| Molar mass | 319.229 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| | |
Ibandronic acid (INN) or ibandronate sodium (USAN) is a potent bisphosphonate drug developed by Hoffman La Roche and used in the prevention and treatment of osteoporosis and metastasis-associated skeletal fractures in people with cancer.[1] It may also be used to treat hypercalcemia (elevated blood calcium levels). Global sales in 2008 were 1.1B CHF ($1.0B USD at 01/01/2009 exchange rates).[2]
Medical uses
Ibandronate is indicated for the treatment and prevention of osteoporosis in post-menopausal women.[3] In May 2003, the U.S. Food and Drug Administration (FDA) approved Ibandronate as a daily treatment for post-menopausal osteoporosis. The basis for this approval was a three-year, randomized, double-blind, placebo-controlled trial women with post-menopausal osteoporosis. Every participant also received daily oral doses of calcium and 400IUs [international units] of vitamin D. At the study's conclusion, both doses significantly reduced the occurrence risk of new vertebral fractures by 50–52 percent when compared to the effects of the placebo drug.
Ibandronate is efficacious for the prevention of metastasis-related bone fractures in multiple myeloma, breast cancer, and certain other cancers.[4]
Adverse effects
In 2008, the U.S Food and Drug Administration (FDA) issued a communication warning of the possibility of severe and sometimes incapacitating bone, joint and/or muscle pain.[5] A study conducted by the American Society of Bone and Mineral Research concluded that long-term use of bisphosphonates, including Boniva, may increase the risk of a rare but serious fracture of the femur. [6]
Brand names
Ibandronic acid is marketed under the trade names Boniva in the USA, Bondronat in Europe, Bonviva in Asia, Ibandrix in Ecuador and Bondrova in Bangladesh.
References
- ↑ Bauss F, Schimmer RC (March 2006). "Ibandronate: the first once-monthly oral bisphosphonate for treatment of postmenopausal osteoporosis". Therapeutics and clinical risk management. 2 (1): 3–18. PMC 1661644
 . PMID 18360577.
. PMID 18360577. - ↑ "Roche 2008 Annual Report" (PDF). Roche. Retrieved 2009-08-13.
- ↑ "boniva". The American Society of Health-System Pharmacists. Retrieved 3 April 2011.
- ↑ Sittig HB (2012). "Pathogenesis and bisphosphonate treatment of skeletal events and bone pain in metastatic cancer: focus on ibandronate". Onkologie. 35 (6): 380–7. doi:10.1159/000338947. PMID 22722461.
- ↑ "Information for Healthcare Professionals: Bisphosphonates (marketed as Actonel, Actonel+Ca, Aredia, Boniva, Didronel, Fosamax, Fosamax+D, Reclast, Skelid, and Zometa)". U.S. Food and Drug Administration. Retrieved 27 October 2010.
- ↑ "Drugs Commonly Prescribed for Osteoporosis Patients are Effective at Reducing Risk of Hip and Spine Fractures, But Panel Says May be Related to Unusual Thigh Bone Fractures When Used Long Term". Journal of Bone and Mineral Research=27 October 2010.
External links
- Ibandronate bound to proteins in the PDB
- Boniva Osteoporosis Treatment by GlaxoSmithKline and Roche Laboratories
- Boniva adverse events reported to the FDA