Turn (biochemistry)
A turn is an element of secondary structure in proteins where the polypeptide chain reverses its overall direction.
Definition
According to one definition,[1] a turn is a structural motif where the Cα atoms of two residues separated by few (usually 1 to 5) peptide bonds are close (< 7 Å), while the residues do not form a secondary structure element such as an alpha helix or beta sheet with regularly repeating backbone dihedral angles. Although the proximity of the terminal Cα atoms usually correlates with formation of a hydrogen bond between the corresponding residues, a hydrogen bond is not a requirement in this turn definition. That said, in many cases the H-bonding and Cα-distance definitions are equivalent.
Types of turns
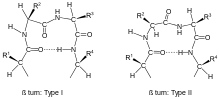
Turns are classified[2] according to the separation between the two end residues:
- In an α-turn the end residues are separated by four peptide bonds ().
- In a β-turn (the most common form), by three bonds ().
- In a γ-turn, by two bonds ().
- In a δ-turn, by one bond () (sterically unlikely).
- In a π-turn, by five bonds ().
| Type | ||||
|---|---|---|---|---|
| I | -60 | -30 | -90 | 0 |
| II | -60 | 120 | 80 | 0 |
| VIII | -60 | -30 | -120 | 120 |
| I' | 60 | 30 | 90 | 0 |
| II' | 60 | -120 | -80 | 0 |
| VIa1 | -60 | 120 | -90 | 0* |
| VIa2 | -120 | 120 | -60 | 0* |
| VIb | -135 | 135 | -75 | 160* |
| IV |
turns excluded from all the above categories | |||
Within each type, turns may be further classified by their backbone dihedral angles (see Ramachandran plot). A turn can be converted into its inverse turn (in which the main chain atoms have opposite chirality) by changing the sign on its dihedral angles. (The inverse turn is not a true enantiomer since the Cα atom chirality is maintained.) Thus, the γ-turn has two forms, a classical form with (φ, ψ) dihedral angles of roughly (75°, -65°) and an inverse form with dihedral angles (-75°, 65°). At least eight forms of the beta turn occur, varying in whether a cis isomer of a peptide bond is involved and on the dihedral angles of the central two residues. The classical and inverse β-turns are distinguished with a prime, e.g., type I and type I' beta turns. If an i->i+3 hydrogen bond is taken as the criterion for turns, the four categories of Venkatachalam[4] (I, II, II', I') suffice[5] to describe all possible beta turns. All four occur frequently in proteins but I is most common, followed by II, I' and II' in that order.
Loops
An ω-loop is a catch-all term for a longer, extended or disordered loop without fixed internal hydrogen bonding.
Multiple turns
In many cases, one or more residues are involved in two partially overlapping turns. For example, in a sequence of 5 residues, both residues 1-4 and residues 2-5 form a turn; in such a case, one speaks of a double turn. Multiple turns (up to 7-fold) occur commonly in proteins.[6] Beta bend ribbons are a different type of multiple turn.
Hairpins
A hairpin is a special case of a turn, in which the direction of the protein backbone reverses and the flanking secondary structure elements interact. For example, a beta hairpin connects two hydrogen-bonded, antiparallel β-strands. (a rather confusing name, since a β-hairpin may contain many types of turns - α,β,γ, etc.)
Beta hairpins may be classified according to the number of residues that make up the turn - that is, that are not part of the flanking β-strands.[7] If this number is X or Y (according to two different definitions of β sheets) the β hairpin is defined as X:Y
Beta turns at the loop ends of beta hairpins have a different distribution of types from the others; type I' is commonest, followed by types II', I and II.
Role in protein folding
Two hypotheses have been proposed for the role of turns in protein folding. In one view, turns play a critical role in folding by bringing together and enabling or allowing interactions between regular secondary structure elements. This view is supported by mutagenesis studies indicating a critical role for particular residues in the turns of some proteins. Also, nonnative isomers of X-Pro peptide bonds in turns can completely block the conformational folding of some proteins. In the opposing view, turns play a passive role in folding. This view is supported by the poor amino-acid conservation observed in most turns. Also, non-native isomers of many X-Pro peptide bonds in turns have little or no effect on folding.
Betaturn Prediction Methods
Over the years, many betaturn prediction methods have been developed. Recently, Dr. Raghava's Group developed BetaTPred3 method which predicts a complete betaturn rather than individual residues falling in betaturn. The method also achieves good accuracy and is the first method which predicts all 9 types of betaturns. Apart from prediction, this method can also be used to find minimum number of mutations required to initiate or break a betaturn in a protein at desired location.
See also
Notes
- ↑ see Rose et al. 1985 in the References
- ↑ Toniolo 1980
- ↑ Venkatachalam 1968; Richardson 1981; Hutchinson and Thornton 1994
- ↑ Venkatachalam, CM (1968). "Sterochemical criteria for polypeptides and proteins. V. Conformations of a system of three linked peptide units.". Biopolymers. 6 (10): 1425–1436. doi:10.1002/bip.1968.360061006. PMID 5685102.
- ↑ Richardson, JS. "The anatomy and taxonomy of protein structure". Adv Protein Chem. 34: 167–339. doi:10.1016/s0065-3233(08)60520-3.
- ↑ Hutchinson 1994, p 2213
- ↑ Sibanda 1989
External links
- BetaTPred3 - Insilico platform for predicting and initiating betaturns in a protein at desired location Article Link
- NetTurnP - Prediction of Beta-turn regions in protein sequences
- BetaTPred - Prediction of Beta Turns in proteins using statistical algorithms
References
These references are ordered by date.
- Venkatachalam CM. (1968). "Stereochemical criteria for polypeptides and proteins. V. Conformation of a system of three linked peptide units". Biopolymers. 6 (10): 1425–36. doi:10.1002/bip.1968.360061006. PMID 5685102.
- Némethy, George; Printz, Morton P. (1972). "The -Turn, a Possible Folded Conformation of the Polypeptide Chain. Comparison with the β-Turn". Macromolecules. 5 (6): 755. doi:10.1021/ma60030a017.
- Lewis PN, Momany FA, Scheraga HA (1973). "Chain reversals in proteins". Biochim Biophys Acta. 303 (2): 211–29. doi:10.1016/0005-2795(73)90350-4. PMID 4351002.
- Toniolo C.; Benedetti, Ettore (1980). "Intramolecularly hydrogen-bonded peptide conformations". CRC Crit Rev Biochem. 9 (1): 1–44. doi:10.3109/10409238009105471. PMID 6254725.
- Richardson JS. (1981). "The anatomy and taxonomy of protein structure". Adv Protein Chem. 34: 167–339. doi:10.1016/S0065-3233(08)60520-3. PMID 7020376.
- Rose GD, Gierasch LM, Smith JA (1985). "Turns in peptides and proteins". Adv Protein Chem. 37: 1–109. doi:10.1016/S0065-3233(08)60063-7. PMID 2865874.
- Milner-White EJ & Poet R (1987). "Loops, bulges, turns and hairpins in proteins". Trends Biochem Sci. 12: 189–192. doi:10.1016/0968-0004(87)90091-0.
- Wilmot CM, Thornton JM (1988). "Analysis and prediction of the different types of beta-turn in proteins". J Mol Biol. 203 (1): 221–32. doi:10.1016/0022-2836(88)90103-9. PMID 3184187.
- Sibanda, B.L.; Blundell, T.L.; Thornton, J.M. (1989). "Conformation of β-hairpins in protein structures:: A systematic classification with applications to modelling by homology, electron density fitting and protein engineering". Journal of Molecular Biology. 206 (4): 759–777. doi:10.1016/0022-2836(89)90583-4. PMID 2500530. Retrieved 2011-02-15.
- Milner-White, E (1990). "Situations of gamma-turns in proteinsTheir relation to alpha-helices, beta-sheets and ligand binding sites". J. Mol. Biol. 216 (2): 385–397. doi:10.1016/S0022-2836(05)80329-8. PMID 2254936.
- Hutchinson, E.G.; Thornton, J.M. (1994). "A revised set of potentials for β-turn formation in proteins". Protein Science. 3 (12): 2207–2216. doi:10.1002/pro.5560031206. PMC 2142776
 . PMID 7756980.
. PMID 7756980. - Pavone V, Gaeta G, Lombardi A, Nastri F, Maglio O, Isernia C, Saviano M (1996). "Discovering protein secondary structures: classification and description of isolated alpha-turns". Biopolymers. 38 (6): 705–21. doi:10.1002/(SICI)1097-0282(199606)38:6<705::AID-BIP3>3.0.CO;2-V. PMID 8652792.
- Rajashankar KR, Ramakumar S (1996). "Pi-turns in proteins and peptides: Classification, conformation, occurrence, hydration and sequence". Protein Sci. 5 (5): 932–46. doi:10.1002/pro.5560050515. PMC 2143406
 . PMID 8732765.
. PMID 8732765.