Myelinogenesis
Myelinogenesis is generally the proliferation of myelin sheaths throughout the nervous system, and specifically the progressive myelination of nerve axon fibers in the central nervous system. This is a non-simultaneous process that occurs primarily postnatally in mammalian species, beginning in the embryo during the midst of early development and finishing after birth.[1]
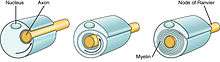
Function
The myelination process allows neuronal signals to propagate down an axon more swiftly without the loss of signal. This enables better connectivity within specific brain regions and also improves broader neuronal pathways connecting spatially separate regions required for many sensory, cognitive, and motor functions.
Some scientists consider myelination to be a key human evolutionary advantage, enabling greater processing speeds that lead to further brain specialization. Myelination continues for at least another 10 to 12 years after birth before an individual is fully developed.[2] While the rate at which individual children develop varies, the sequence of development is the same for all children (with a range of ages for specific developmental tasks to take place).
Stages
Oligodendrocytes are responsible for the creation of myelin sheaths. There are “two stages of OL markers, differentiation of OPCs to OLs, and ensheathment of axons…”.[3]
Although the mechanisms and processes of myelination are yet to be fully understood, some specific stages in this process have become clear:
- Stage 1: Axon contact
- Stage 2: Glial cell gene production
- Stage 3: Axon ensheathment, which is one of two phases in the early stages of the formation of myelin sheath. Spiral ensheathment of target axons begins through the elaboration from each initiator process of lamellar extensions which extend circumferentially around the target axon and thereby form the first turn of its myelin sheath.[4]
- Stage 4: Maturation
.svg.png)
Mechanism and process
The principal molecular mechanisms that control the process and sequence of myelinogenesis are not entirely known. Numerous studies have primarily focused on simplifying the underlying neuronal control of myelinogenesis and such studies have provided several possibilities.
One early study focused on the signaling of oligodendrocyte myelination by regenerating peripheral axons. Researchers studied regenerating PNS axons for 28 weeks in order to investigate whether or not peripheral axons stimulate oligodendrocytes to begin myelination. Experimental induction of myelination by regenerating peripheral axons demonstrated that Schwann cells and oligodendrocytes have a shared mechanism to stimulate myelination.[5] A similar study working to provide evidence for neuronal regulation of myelinogenesis suggested that myelin formation was due to Schwann cells that were controlled by an undefined property of an associated axon.[5]
Another such study in mice determined that the helix-loop-helix transcription factor, OLIG1, plays an integral role in the process of oligodendrocyte myelinogenesis. OLIG1 controls regulation in several myelin related genes, while suppressing others. On a cellular level, the study experimentally demonstrated that OLIG1 is necessary in order to stimulate myelination by oligodendrocytes in the brain. However, spinal cord related oligodendrocytes demonstrated a significantly smaller need of OLIG1 regulation in order to begin myelination.[6]
Recent research in rats has suggested that the separate action of apotransferrin and thyroid hormone could have an important role in myelination. Moreover, research also seems to indicate that the function of transferrin and thyroid hormone together act to control myelinogenesis.
Primarily, this research indicated that due to hyperthyroidism that resulted from an increase in transferrin expression, as well as apotransferrin-dependent regulation of thyroid hormone receptor alpha, meant that transferrin was likely related to thyroid hormone’s effects on oligodendrocyte maturation, and eventually myelination.
The relationship is believed to take place at the thyroid hormone receptor level. Immunohistochemistry analysis was utilized in order to further confirm the relationship between thyroid hormone and apotransferrin during oligodendrogenesis.[7]
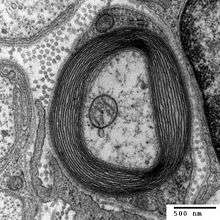
Studies on the control of myelinogenesis
Studies have revealed that myelinogenesis is controlled by the synthesis of proteins P1, P2, and P0.[8] By using SDS-PAGE, researchers revealed distinct bands with band sizes of 27,000 Daltons (P1), 19,000 Daltons (P2), and 14,000 Daltons (P0). Studies have also shown that P1 and P2 are active before Po since this protein comes from the peripheral nervous system.[8] In the process of regeneration, Schwann cells re-synthesize proteins associated with myelin-specific proteins when axonal presence is re-established. A key part of the experiment was to indicate that Schwann cells don't synthesize myelin-proteins in the absence of axons. Synthesis of detectable myelin-specific proteins did not occur in Schwann cells free of axons and thus proving that the axons instruct the Schwann cells to initiate the production of myelin proteins immediately after establishment of axonal association.[8]
Even though the axon completely controlled the synthesis of these proteins, the axon alone isn’t enough for myelinogenesis. As the experiment progressed, it became more evident that membrane-membrane interactions between axons somehow promoted the synthesis of the essential proteins P1, P2, and P0. It is likely that axons from myelinated fibers are able to induce Schwann cell myelin protein synthesis throughout the adult life.
Myelinogenesis in the optic nerve
The process and mechanistic function of myelinogenesis has traditionally been studied using ultrastructure and biochemical techniques in rat optic nerves. The implementation of this method of study has long allowed for experimental observation of myelinogenesis in a model organism nerve that consists entirely of unmyelinated axons. Furthermore, the use of the rat optic nerve helped provide insight for early myelinogenesis researchers into improper and atypical courses of myelinogenesis.[9]
One early study showed that in the developing rat optic nerves, formation of oligodendrocytes and subsequent myelination occurs postnatal. In the optic nerve, the oligodendrocyte cells divided for the final time at five days, with the onset of myelin formation occurring on or around day 6 or 7. However, the exact process by which the oligodendrocytes were stimulated to produce myelin was not yet fully understood, but early myelination in the optic nerve has been linked to a rise in the production of various lipids – cholesterol, cerebroside, and sulfatide.[9]
As researchers began to do postnatal research, they found that myelinogenesis in the rat optic nerve initially commences with axons the largest diameters before proceeding to the remaining smaller axons. In the second week postnatal, oligodendrocyte formation slowed – at this point, 15% of axons have been myelinated – however, myelinogenesis continued to rapidly increase. During the fourth week postnatal, nearly 85% of the axons in the rat optic had been myelinated.[9] During the fifth week and onward toward week sixteen, the myelination decelerated and the remaining unmyelinated axons were ensheathed in myelin.[10] Through the rat optic nerve, early research made significant contributions to knowledge in the field of myelinogenesis.

Importance of sulfate
Studies on the developing optic nerve revealed that galactocerebroside (which forms sulfatide) appeared on the 9th post-natal day and reached a peak on the 15th post-natal day.[9] This expression was similar to a period where the optic nerve showed a maximal myelination period of the axon. As the activity of axon myelination decreased, and one could conclude that the activity of the enzyme is paralleled with the incorporation of sulfate ([35S]) into sulfatide in vivo.
The studies on a rat optic nerve revealed that 15 days post-natal is when an increase in myelination is observed. Before this time period, most of the axons, roughly about 70%, are not myelinated. At this time, [35S] Sulfate was incorporated into sulfatide and the activity of cerebroside, sulfotransferase reached a peak in enzyme activity. This time frame also showed a period of maximal myelination based on the biochemical data.[9]
In the Central Nervous System, sulfatide, sulfated glycoproteins, and sulfated mucopolysaccharides appear to be associated with neurons rather than myelin. When graphing the amount of sulfatide made from [35S] and the activity of sulfotransferase, we get to distinguished peaks.[9] The peaks occur on the 15th post-natal day. These peaks corresponded with the maximal myelination period of the optic nerve that has been seen throughout the experiment.[9]
In conclusion, the early phase of myelination was correlated with the increases synthesis of lipids, cholesterol, cerebroside, and sulfatide.[9] It is likely that these compounds are synthesized and packaged in the Golgi Apparatus of oligodendroglia.[9] Even though the transport of these lipids is unknown, it appears that myelination is delayed without their synthesis.
Clinical significance
Myelin is an insulating material that forms the myelin sheath and wraps around the axon of the neuron to allow functioning in the nervous system.
One disease that has significance that affects Myelinogenesis is Multiple Sclerosis. Multiple sclerosis (MS) is a demyelinating disease of the central nervous system (CNS).[11] A symptom of multiple sclerosis is demyelination.There has been tests to find a treatment that can potentially slow down or prevent demyelination but there have been none. However, there are possible strategies that may potentially "remyelinate" myelin sheaths in MS.
Oligodendrocytes are a certain type of cell that functions to generate myelin sheath. Recent work on this cell has provided needed insight on the rolling processes: the process of demyelination, the spontaneous ability of the CNS to regenerate myelin, and the inevitable failure of "remyelination".[11] Studies that focused on oligodendrocytes made it possible to look at different molecular targets that could allow cells to be protected from demyelination and allow remyelination. Combining immunomodulatory therapy with strategies to protect oligodendrocytes from further degeneration and enhance remyelination present a very real means to improve clinical outcomes for chronic progressive patients in the near future.[11]
The mechanisms targeted by the researchers were: 1) halting the pathologic immune response, 2) protecting the CNS from further damage, and 3) repairing the damage via regeneration of myelin sheaths, with the overarching goals being to restore conduction and prevent from further axonal loss.[11]
There are drugs that would relieve the symptoms of MS but it will not cure or stop the progression of MS. MS can progressively get worse overtime left untreated. With some of the approaches discovered, it may be possible to protect oligodendrocytes from degenerating and enhance regeneration with immunotherapies.[11]
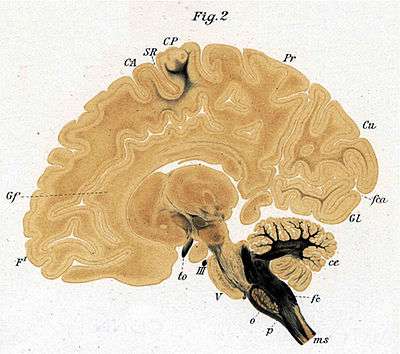
Research History
Another researcher, Paul Flechsig spent most of his career studying and publishing the details of the process in the cerebral cortex of humans. This takes place mostly between two months before and after birth. He identified 45 separate cortical areas and, in fact, mapped the cerebral cortex by the myelination pattern. The first cortical region to myelinate is in the motor cortex (part of Brodmann's area 4), the second is the olfactory cortex and the third is part of the somatosensory cortex (BA 3,1,2).
The last areas to myelinate are the anterior cingulate cortex (F#43), the inferior temporal cortex (F#44) and the dorsolateral prefrontal cortex (F#45).
In the cerebral convolutions, as in all other parts of the central nervous system, the nerve-fibres do not develop everywhere simultaneously, but step by step in a definite succession, this order of events being particularly maintained in regard to the appearance of the medullary substance. In the convolutions of the cerebrum the investment with medullary substance (myelinisation) has already begun in some places three months before the maturity of the foetus, whilst in other places numerous fibres are devoid of medullary substance even three months after birth. The order of succession in the convolutions is governed by a law identical with the law which I have shown holds good for the spinal cord, the medulla oblongata, and the mesocephalon, and which may be stated somewhat in this way- that, speaking approximately, equally important nerve-fibres are developed simultaneously, but those of dissimilar importance are developed one after another in a succession defined by an imperative law (Fundamental Law of Myelogenesis). The formation of medullary substance is almost completed in certain convolutions at a time when in some it is not even begun and in others has made only slight progress.[12]
References
- ↑ Eilam, R.; Bar-Lev, D.D.; Levin-Zaidman, S.; Tsoory, M.; LoPresti, P.; Sela, M.; Arnon, R.; Aharoni, R. "Oligodendrogenesis and meylinogenesis during postnatal development effect of glatiramer acetate". Glia. 62: 649-665. doi:10.1002/glia.22632.
- ↑ Paus, T., Zijdenbos, A., Worsley, K., Collins, D. L., Blumenthal, J., Giedd, J. N., ... & Evans, A. C. (1999). Structural maturation of neural pathways in children and adolescents: in vivo study. Science, 283(5409), 1908-1911.
- ↑ Watkins, T., Mulinyawe, S., Emery, B., Barres, B. (2008). Distinct Stages of Myelination Regulated by Y-Secretase and Astrocytes in a Rapidly Myelinating CNS Coculture System. 555-569
- ↑ Friedrich, VL., Hardy, RJ., (1996). Progressive Remodeling of the Oligodendrocyte Process Arbor during Myelinogenesis. 243-54.
- 1 2 Weinberg, E., & Spencer, P. (1979). Studies on the control of myelinogenesis. 3. Signaling of oligodendrocyte myelination by regenerating peripheral axons. Brain Research, 162(2), 273-279. doi:10.1016/0006-8993(79)90289-0
- ↑ Xin, M. (2005). Myelinogenesis and Axonal Recognition by Oligodendrocytes in Brain Are Uncoupled in Olig1-Null Mice. Journal Of Neuroscience, 25(6), 1354-1365. doi:10.1523/jneurosci.3034-04.2005
- ↑ Marziali, L.N., Garcia, C.I., Pasquini, J.M. (2015). Transferrin and thyroid hormone converge in the control of myelinogenesis. Experimental Neurology. Vol 265. 129–141.
- 1 2 3 Politis, MJ, N. Sternberger, Kathy Ederle, and Peter S. Spencer. "Studies on the Control of Myelinogenesis." The Journal of Neuroscience 2.9 (1982): 1252-266.
- 1 2 3 4 5 6 7 8 9 Tennekoon, GI., Cohen, SR., Price, DL., McKhann, GM. (1977). Myelinogenesis in optic nerve. A morphological, autoradiographic, and biochemical analysis. Journal Of Cell Biology, 72(3), 604-616.
- ↑ Dangata, Y., Kaufman, M. (1997). Myelinogenesis in the Optic Nerve of (C57BL x CBA) F1 Hybrid Mice: A Morphometric Analysis.European Journal Of Morphology, 35(1), 3-18.
- 1 2 3 4 5 Rodgers, Jane M.; Robinson, Andrew P.; Miller, Stephen D. (2013). "Strategies for protecting oligodendrocytes and enhancing remyelination in multiple sclerosis". Discover Medicine. 86: 53-63.
- ↑ Flechsig, Paul (1901-10-19). "Developmental (myelogenetic) localisation of the cerebral cortex in the human subject". The Lancet: 1028. doi:10.1016/s0140-6736(01)01429-5.