Prochirality
In stereochemistry, prochiral molecules are those that can be converted from achiral to chiral in a single step.[1][2] An achiral species which can be converted to a chiral in two steps is called proprochiral.[2]
If two identical substituents are attached to a sp3-hybridized atom, the descriptors pro-R and pro-S are used to distinguish between the two. Promoting the pro-R substituent to higher priority than the other identical substituent results in an R chirality center at the original sp3-hybridized atom, and analogously for the pro-S substituent.
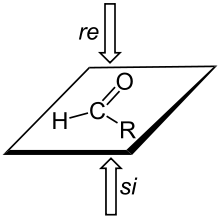
A trigonal planar sp2-hybridized atom can be converted to a chiral center when a substituent is added to the re or si face of the molecule (pronounced "ray" and "sigh"). A face is labeled re if, when looking at that face, the substituents at the trigonal atom are arranged in decreasing Cahn-Ingold-Prelog priority order in a clockwise order, and si if the priorities decrease in counter-clockwise order; note that the designation of the resulting chiral center as S or R depends on the priority of the incoming group.
References
- ↑ John McMurry. Organic Chemistry (6ed ed.). Brooks/Cole. pp. 301–303.
- 1 2 IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "prochirality".