Renalase
| RNLS | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||
| Identifiers | |||||||||||||||||
| Aliases | RNLS, C10orf59, RENALASE, renalase, FAD-dependent amine oxidase, renalase, FAD dependent amine oxidase | ||||||||||||||||
| External IDs | MGI: 1915045 HomoloGene: 41254 GeneCards: RNLS | ||||||||||||||||
| |||||||||||||||||
| Orthologs | |||||||||||||||||
| Species | Human | Mouse | |||||||||||||||
| Entrez | |||||||||||||||||
| Ensembl | |||||||||||||||||
| UniProt | |||||||||||||||||
| RefSeq (mRNA) | |||||||||||||||||
| RefSeq (protein) | |||||||||||||||||
| Location (UCSC) | Chr 10: 88.27 – 88.58 Mb | Chr 19: 33.14 – 33.39 Mb | |||||||||||||||
| PubMed search | [1] | [2] | |||||||||||||||
| Wikidata | |||||||||||||||||
| View/Edit Human | View/Edit Mouse |
Renalase, FAD-dependent amine oxidase is an enzyme that in humans is encoded by the RNLS gene. Renalase is a flavin adenine dinucleotide-dependent amine oxidase that is secreted into the blood from the kidney.[3]
Structure
Gene
The gene encoding this protein is called RNLS (also known as C10orf59 or FLJ11218).[3] The renalase gene has 9 exons spanning approximately 311,000 bp and resides on chromosome 10 at q23.33.[4]
Protein
The renalase protein consists of a putative secretory signal peptide (SignalP score of 0.4), a flavin adenine dinucleotide (FAD)-binding region, and an oxidase domain. At least four alternative splicing isoforms have been identified in humans (hRenalase1 to hRenalase4). Only hRenalase1 is detected in human blood samples, which means that hRenalase2 to 4 probably have different functions than hRenalase1.[5]
Analysis of the primary structure of renalase shows that it is an FAD-dependent oxidase. The X-ray crystal structure of hRenalase1 reveals structural similarity between renalase and p-hydroxybenzoate hydroxylase.[6]
Function
Renalase has been claimed to degrade catecholamines like adrenaline (epinephrine) and noradrenaline (norepinephrine) in the blood circulation.[5] Dr. Gary Desir's laboratory at Yale School of Medicine discovered and named renalase in 2005[4] suggest that the human kidney releases this protein into the bloodstream to regulate blood pressure (in addition to other possible, as yet undiscovered, functions).[5]
Whether renalase actually oxidizes catecholamine substrates has been widely disputed.[7][8] The primary evidence for catecholamine oxidation is the detection of H2O2, however catecholamines emanate H2O2 in the presence of O2 in a natural decomposition reaction. In 2013, renalase was claimed to oxidize α-NADH (the normal form of NADH is the β anomer) to β-NAD+, with concomitant reduction of O2 (dioxygen) to H2O2 (hydrogen peroxide).[9] This reaction was proposed to repair aberrant NADH and NADPH forms that are not accepted as cofactors by most nicotinamide-dependent oxidoreductase enzymes.
It transpired that α-NAD(P)H molecules are not substrates for renalase; instead 6-dihydroNAD (6DHNAD) was identified as the substrate, a molecule with highly similar spectrophotometric characteristics and equilibrium concentrations as those reported for α-NAD(P)H.[10] 6DHNAD is an isomeric form of β-NADH that carries the hydride in the 6-position of the nicotinamide base as opposed to the metabolically active 4-position. This form of NAD is one of three products that are formed from non-enzymatic reduction of β-NAD+ in addition to 4-dihydroNAD (β-NADH), 2-dihydroNAD (2DHNAD). Both 2DHNAD and 6DHNAD were shown to be substrates for renalase. These molecules react rapidly to reduce the enzyme's flavin cofactor forming β-NAD+. The renalase flavin then delivers the electrons harvested to O2 (dioxygen) forming H2O2 (hydrogen peroxide), completing the catalytic cycle. It was shown that both 6DHNAD and 2DHNAD are tight binding inhibitors of specific primary metabolism dehydrogenases, thereby defining a clear metabolic function for renalase in the alleviation of this inhibition.
Extracellular renalase functions as a survival and growth factor, independent of its enzymatic activity.[11] Either naturally folded renalase or a 20 amino acid renalase peptide can activate the phosphoinositide 3-kinase (PI3K)and the mitogen-activated protein kinase (MAPK) pathways in a manner that protects cells against apoptosis.
Catalysis
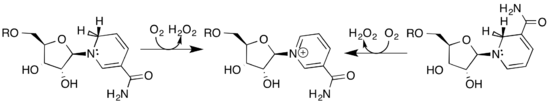
Renalase isolated in native form, that is, without refolding steps, catalyzes the oxidation of 6DHNAD(P) or 2DHNAD(P), the isomeric forms of β-NAD(P)H .[9] In contrast to clear evidence for catalysis of this activity, the native renalase used in these experiments did not catalyze the conversion of the catecholamine epinephrine to adrenochrome.[9]
Renalase is secreted in plasma, and functions as an anti-apoptotic survival factor. The Plasma membrane Ca2+ ATPase PMCA4b is a putative receptor for extracellular renalase.[12] The binding of renalase to PMCA4b stimulates calcium efflux with subsequent activation of the PI3K and MAPK pathways, increased expression of the anti-apoptotic factor Bcl-2 , and decreased caspase3-mediated apoptosis. Administration of recombinant renalase protects against acute kidney injury (AKI), and against cardiac ischemia in animal models.
Clinical significance
Renalase levels are markedly reduced in patients with severe chronic kidney disease (end-stage renal disease, ESRD). Since hormones like erythropoietin are secreted less in ESRD, renalase may also be a kidney hormone, although it is also expressed in heart muscle, skeletal muscle and liver cells in humans, and in mouse testicles.[5][13]
Renalase has been controversially proposed to degrade catecholamines, which are hormones involved in the acute stress (fight-or-flight) response. Injection of renalase in rodents transiently decreases blood pressure, heart rate, heart muscle contractility, and blood vessel resistance.[4] Under normal conditions, renalase is present but inactive in the bloodstream. When catecholamines are released into the bloodstream however, renalase activity increases about tenfold within 30 seconds, and remains high for an hour or longer. Activation of circulating renalase is probably responsible for early activation, while secretion into the bloodstream occurs after 15 minutes.[14]
polymorphisms in the renalase gene is a risk factor for essential hypertension.[15]
Single nucleotide polymorphisms in the renalase gene are associated with type 1 diabetes. A genome-wide association study and meta-analysis found that approximately 42 loci affect the risk of diabetes[16] The data confirmed linkage with most of the 24 previously identified loci, and identified 27 novel loci. The strongest evidence of association among these novel regions was achieved for the renalase gene. Renalase gene polymorphism is associated with increased blood pressure in preeclampsia. doi:10.1016/j.preghy.2016.04.002
Animal studies
In mice, hearts exposed to oxygen shortage (ischemia), myocardial infarction size was decreased and heart function better preserved when renalase was administered.[17] Renalase knock-out mice are also more sensitive to damage to the heart muscle.[5] Renalase expression in the heart is also decreased in the rat model of end-stage renal disease. The scientists who discovered renalase believe that it might explain some of the susceptibility to heart disease among patients with chronic kidney disease.[5]
Using small inhibitory RNAs or knock-out mice, the consequences of loss of renalase function have been studied. These include raised blood pressure (hypertension), increased heart rate (tachycardia), increased blood vessel resistance (vasoconstriction) and an increased catecholamine response.[18][19]
In a rat model of chronic kidney disease (in which 85% of kidney tissue is surgically removed), renalase deficiency and defective renalase activation develops 2–3 weeks after surgery.[14]
Interactions
Renalase has been shown to interact with PMCA4b.[20]
References
- ↑ "Human PubMed Reference:".
- ↑ "Mouse PubMed Reference:".
- 1 2 "Entrez Gene: Renalase, FAD-dependent amine oxidase".
- 1 2 3 Xu J, Li G, Wang P, Velazquez H, Yao X, Li Y, Wu Y, Peixoto A, Crowley S, Desir GV (May 2005). "Renalase is a novel, soluble monoamine oxidase that is reported to regulate cardiac function and blood pressure". The Journal of Clinical Investigation. 115 (5): 1275–80. doi:10.1172/JCI24066. PMC 1074681
 . PMID 15841207.
. PMID 15841207. - 1 2 3 4 5 6 Desir GV (Aug 2009). "Regulation of blood pressure and cardiovascular function by renalase". Kidney International. 76 (4): 366–70. doi:10.1038/ki.2009.169. PMID 19471322.
- ↑ Milani M, Ciriello F, Baroni S, Pandini V, Canevari G, Bolognesi M, Aliverti A (Aug 2011). "FAD-binding site and NADP reactivity in human renalase: a new enzyme involved in blood pressure regulation". Journal of Molecular Biology. 411 (2): 463–473. doi:10.1016/j.jmb.2011.06.010. PMID 21699903.
- ↑ Boomsma F, Tipton KF (2007). "Renalase, a catecholamine-metabolising enzyme?". Journal of Neural Transmission (Vienna, Austria : 1996). 114 (6): 775–6. doi:10.1007/s00702-007-0672-1. PMC 2793395
 . PMID 17385068.
. PMID 17385068. - ↑ Baroni S, Milani M, Pandini V, Pavesi G, Horner D, Aliverti A (2013). "Is renalase a novel player in catecholaminergic signaling? The mystery of the catalytic activity of an intriguing new flavoenzyme". Current Pharmaceutical Design. 19 (14): 2540–2551. doi:10.2174/1381612811319140005. PMID 23116393.
- 1 2 3 Beaupre BA, Carmichael BR, Hoag MR, Shah DD, Moran GR (Sep 2013). "Renalase is an α-NAD(P)H oxidase/anomerase". Journal of the American Chemical Society. 135 (37): 13980–13987. doi:10.1021/ja407384h. PMID 23964689.
- ↑ Beaupre BA, Hoag MR, Roman J, Försterling FH, Moran GR (Jan 2015). "Metabolic Function for Human Renalase: Oxidation of Isomeric Forms of β-NAD(P)H that Are Inhibitory to Primary Metabolism". Biochemistry. 54 (3): 795–806. doi:10.1021/bi5013436. PMID 25531177.
- ↑ Wang L, Velazquez H, Moeckel G, Chang J, Ham A, Lee HT, Safirstein R, Desir GV (Jun 2014). "Renalase prevents AKI independent of amine oxidase activity". Journal of the American Society of Nephrology : JASN. 25 (6): 1226–1235. doi:10.1681/ASN.2013060665. PMID 24511138.
- ↑ Desir GV, Wang L, Velazquez H, Chang J, Safirstein R (2014). "Identification of a receptor for extracellular renalase". J Am Soc Nephrol. 25: 384A.
- ↑ Wang J, Qi S, Cheng W, Li L, Wang F, Li YZ, Zhang SP (Dec 2008). "Identification, expression and tissue distribution of a renalase homologue from mouse". Molecular Biology Reports. 35 (4): 613–20. doi:10.1007/s11033-007-9131-1. PMID 17846919.
- 1 2 Li G, Xu J, Wang P, Velazquez H, Li Y, Wu Y, Desir GV (Mar 2008). "Catecholamines regulate the activity, secretion, and synthesis of renalase". Circulation. 117 (10): 1277–82. doi:10.1161/CIRCULATIONAHA.107.732032. PMID 18299506.
- ↑ Zhao Q, Fan Z, He J, Chen S, Li H, Zhang P, Wang L, Hu D, Huang J, Qiang B, Gu D (Aug 2007). "Renalase gene is a novel susceptibility gene for essential hypertension: a two-stage association study in northern Han Chinese population". Journal of Molecular Medicine (Berlin, Germany). 85 (8): 877–85. doi:10.1007/s00109-006-0151-4. PMID 17216203.
- ↑ Barrett JC, Clayton DG, Concannon P, Akolkar B, Cooper JD, Erlich HA, Julier C, Morahan G, Nerup J, Nierras C, Plagnol V, Pociot F, Schuilenburg H, Smyth DJ, Stevens H, Todd JA, Walker NM, Rich SS (2009). "Genome-wide association study and meta-analysis find that over 40 loci affect risk of type 1 diabetes". Nat. Genet. 41 (6): 703–7. doi:10.1038/ng.381. PMC 2889014
 . PMID 19430480.
. PMID 19430480. - ↑ Desir GV, Li Y, Liu D (2007). "Downregulation of cardiac renalase expression in CKD, and protective effect of renalase in acute coronary syndrome.". J Am Soc Nephrol. 18: 149A.
- ↑ Ghosh SS, Gehr TW, Sica DA, Masilamani S, Ghosh S, Wang R, McGuire E (2006). "Effect of renalase inhibition on blood pressure". J Am Soc Nephrology. 17: 208A.
- ↑ Desir GV, Wu Y, Wang P, et al. (2008). "Renalase deficiency increases sympathetic tone and causes hypertension.". J Am Soc Nephrol.
- ↑ Wang L, Velazquez H, Chang J, Safirstein R, Desir GV (2015). "Identification of a receptor for extracellular renalase". PLOS ONE. 10 (4): e0122932. doi:10.1371/journal.pone.0122932. PMC 4407985
 . PMID 25906147.
. PMID 25906147.