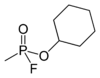
Cyclosarin
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Cyclohexyl methylphosphonofluoridate | |||
| Other names
GF;(Fluoro-methyl-phosphoryl)oxycyclohexane | |||
| Identifiers | |||
| 329-99-7 | |||
| 3D model (Jmol) | Interactive image | ||
| ChemSpider | 58069 | ||
| |||
| |||
| Properties | |||
| C7H14FO2P | |||
| Molar mass | 180.16 g·mol−1 | ||
| Appearance | Colorless liquid | ||
| Density | 1.1278 g/cm3 | ||
| Melting point | −30 °C (−22 °F; 243 K) | ||
| Boiling point | 239 °C (462 °F; 512 K) | ||
| Almost insoluble | |||
| Hazards | |||
| Flash point | 94 °C (201 °F; 367 K) | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
Cyclosarin or GF (cyclohexyl methylphosphonofluoridate) is an extremely toxic substance used as a chemical weapon. It is a member of the G-series family of nerve agents, a group of chemical weapons discovered and synthesized by a German team led by Dr. Gerhard Schrader. The major nerve gases are the G agents, sarin (GB), soman (GD), tabun (GA), and the V agents such as VX. The original agent, tabun, was discovered in Germany in 1936 in the process of work on organophosphorus insecticides. Next came sarin, soman and finally the most toxic, cyclosarin, a product of commercial insecticide laboratories prior to World War II.
As a chemical weapon, it is classified as a weapon of mass destruction by the United Nations. Pursuant to UN Resolution 687 its production and stockpiling was outlawed globally by the Chemical Weapons Convention (CWC) of 1993, although Egypt, Israel, North Korea and South Sudan have not ratified the CWC (thus not outlawing their own stockpiling of chemical weapons).
Chemical characteristics
Like its predecessor sarin, cyclosarin is a liquid organophosphate nerve agent. Its physical characteristics are, however, quite different from sarin.
At room temperature, cyclosarin is a colorless liquid whose odor has been variously described as sweet and musty, or resembling peaches or shellac. Unlike sarin, cyclosarin is a persistent liquid, meaning that it has a low vapor pressure and therefore evaporates relatively slowly, about 69 times slower than sarin and 20 times slower than water.
Also unlike sarin, cyclosarin is flammable, with a flash point of 94 °C (201 °F).
Cyclosarin (GF) also demonstrates greater toxicity than sarin (GB) in humans. Sarin has a median lethal dose (LD50) of 5 mg (for a 70 kg human), while GF has an LD50 of 1.2 mg. The median lethal concentration and time (LCt50) of cyclosarin is 50 mg⋅min/m3, which is half that of GB.
History
First synthesized during World War II as part of Nazi Germany's chemical weapons research on organophosphate compounds after their military potential was discovered, cyclosarin was also studied later in the United States and Great Britain in the early 1950s as part of a systematic study of potential nerve agents. It was never selected for mass production, however, due to its precursors being more expensive than those of other G-series nerve agents such as sarin (GB).
To date, Iraq is the only nation known to have manufactured significant quantities of cyclosarin for use as a chemical agent and to deploy it in battle. During the Iran–Iraq war (1980–1988), the Iraqis used sarin and cyclosarin together as a mixture. This was likely done to obtain a more persistent chemical agent as well as in response to an existing embargo placed on alcohol precursors for sarin.[1]
Munitions
Binary weapons
Like other nerve agents, cyclosarin can be shipped in binary munitions.
A cyclosarin binary weapon would most likely contain methylphosphonyl difluoride in one capsule, with the other capsule containing either cyclohexanol or a mixture of cyclohexylamine and cyclohexanol.
GB-GF mixtures
Iraq fielded munitions filled with a mixture of GB (sarin) and GF (cyclosarin). Tests on mice indicated that GB-GF mixtures have a relative toxicity between GF and GB.[2]
In popular culture
Cyclosarin was central to the plot of the television episode "Chuck Versus the Mask" (Chuck, season 3, episode 7).
References
- United States Central Intelligence Agency. (July 15, 1996). Stability of Iraq's Chemical Weapon Stockpile Retrieved October 30, 2004
- Office of the Special Assistant for Gulf War Illnesses. (Oct. 19, 2004). Chemical Properties of Sarin and Cyclosarin Retrieved October 30, 2004
- Press release from Centcom confirming that the chemical munitions found by the Poles dated back to before the 1991 Gulf War, and, thus, could not represent a threat.
- A laboratory history of chemical warfare agents (2006) edition 2 Jared Ledgard.
- ↑ "Nerve Agent:GF". Retrieved January 5, 2009.
- ↑ "CBWInfo.com". Retrieved March 1, 2012.