Xylylene
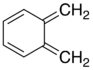
Xylylene comprises two isomeric organic compounds with the formula C6H4(CH2)2. These compounds are related to the corresponding quinones by replacement of the oxygen atoms by CH2 groups. ortho- and para-xylylene are best known, although neither is stable in solid or liquid form. Certain substituted derivatives of xylylenes are however highly stable, an example being tetracyanoquinodimethane.
Synthesis and reactivity
p-Xylylene forms upon pyrolysis of p-xylene or, more readily, the α-substituted derivatives (see equation). Upon condensation, p-xylylene dimerizes with moderate efficiency to give p-cyclophane:[1]
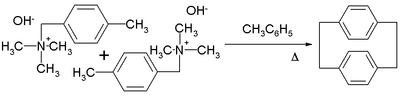
Further heating of the p-cyclophane gives poly(para-xylylene).
The reaction of α,α'-dibromo-o-xylene with iron carbonyls affords low yields of the xylylene complex Fe(CO)3[η4-C6H4(CH2)2]. This complex is similar to Fe(CO)3[η4-1,3-butadiene].[2]
At high temperatures, benzocyclobutenes can undergo electrocyclic ring-opening to form o-xylylenes. This and other syntheses of o-xylylenes, and their subsequent dimerization by [4+4] cycloaddition to form cycloctyl structures, were used repeatedly in the synthesis of superphane.[3]
Electronic structure
Despite the observed chemistry of para-xylylene (i.e. its rapid polymerization to poly-p-xylylene), which suggests the compound exists as a diradical, physical evidence unanimously concludes that the lowest electronic state of p-xylylene is a closed shell singlet. Additionally, several computational methods confirm this assignment.[4]
References
- ↑ H. E. Winberg, F. S. Fawcett "[2.2]Paracyclophane" Organic Syntheses, Coll. Vol. 5, p.883 (1973); Vol. 42, p.83 (1962) Link.
- ↑ R. C. Kerber, E. C. Ribakove "Formation of iron carbonyl complexes of reactive polyenes from dihalides involving the free polyene" Organometallics, 1991, volume 10, pp 2848–2853.doi:10.1021/om00054a059
- ↑ Sekine, Y.; Brown, M.; Boekelheide, V. (1979). "[2.2.2.2.2.2](1,2,3,4,5,6)Cyclophane: superphane". Journal of the American Chemical Society. 101: 3126–3127. doi:10.1021/ja00505a053.
- ↑ Montgomery, L. K., Huffman, J. C., Jurczak, E. A. & Grendze, M. P. The molecular structures of Thiele’s and Chichibabin’s hydrocarbons. J. Am. Chem. Soc. 108, 6004–6011 (1986) DOI:10.1021/ja00279a056