BK channel
| KCNMA1 | |
|---|---|
 The domain structure of BK channels | |
| Identifiers | |
| Symbol | KCNMA1 |
| Alt. symbols | SLO |
| Entrez | 3778 |
| HUGO | 6284 |
| OMIM | 600150 |
| RefSeq | NM_002247 |
| UniProt | Q12791 |
| Other data | |
| Locus | Chr. 10 q22 |
| KCNMB1 | |
|---|---|
| Identifiers | |
| Symbol | KCNMB1 |
| Entrez | 3779 |
| HUGO | 6285 |
| OMIM | 603951 |
| RefSeq | NM_004137 |
| UniProt | Q16558 |
| Other data | |
| Locus | Chr. 5 q34 |
| KCNMB2 | |
|---|---|
| Identifiers | |
| Symbol | KCNMB2 |
| Entrez | 10242 |
| HUGO | 6286 |
| OMIM | 605214 |
| RefSeq | NM_181361 |
| UniProt | Q9Y691 |
| Other data | |
| Locus | Chr. 3 q26.32 |
| KCNMB3 | |
|---|---|
| Identifiers | |
| Symbol | KCNMB3 |
| Alt. symbols | KCNMB2, KCNMBL |
| Entrez | 27094 |
| HUGO | 6287 |
| OMIM | 605222 |
| RefSeq | NM_171828 |
| UniProt | Q9NPA1 |
| Other data | |
| Locus | Chr. 3 q26.3-q27 |
| KCNMB3L | |
|---|---|
| Identifiers | |
| Symbol | KCNMB3L |
| Alt. symbols | KCNMB2L, KCNMBLP |
| Entrez | 27093 |
| HUGO | 6288 |
| RefSeq | NG_002679 |
| Other data | |
| Locus | Chr. 22 q11.1 |
| KCNMB4 | |
|---|---|
| Identifiers | |
| Symbol | KCNMB4 |
| Entrez | 27345 |
| HUGO | 6289 |
| OMIM | 605223 |
| RefSeq | NM_014505 |
| UniProt | Q86W47 |
| Other data | |
| Locus | Chr. 12 q15 |
| Calcium-activated BK potassium channel alpha subunit | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | BK_channel_a | ||||||||
| Pfam | PF03493 | ||||||||
| InterPro | IPR003929 | ||||||||
| |||||||||
BK channels (Big Potassium), also called Maxi-K or slo1, are potassium channels characterized by their large conductance for potassium ions (K+) through cell membranes. These channels are activated (opened) by changes in membrane electrical potential and/or by increases in concentration of intracellular calcium ion (Ca2+).[1][2] Opening of BK channels allows K+ to passively flow through the channel, down the electrochemical gradient. Under typical physiological conditions, this results in an efflux of K+ from the cell, which leads to cell membrane hyperpolarization (an increase in the electrical potential across the cell membrane) and a decrease in cell excitability (a decrease in the probability that the cell will transmit an action potential).[3]
BK channels are essential for the regulation of several key physiological processes including smooth muscle tone and neuronal excitability.[4] They control the contraction of smooth muscle and are involved with the electrical tuning of hair cells in the cochlea. BK channels also contribute to the behavioral effects of ethanol in the worm C. elegans under high exogenous doses (> 100 mM) [5] that have been shown to correspond to biologically relevant internal ethanol concentrations.[6] It remains to be determined if BK channels contribute to intoxication in humans.
Structure
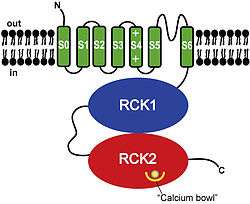
As with most other voltage-gated potassium channels, BK channels have a tetrameric structure. Each monomer of the channel-forming alpha subunit is the product of the KCNMA1 gene. Modulatory beta subunits (encoded by KCNMB1, KCNMB2, KCNMB3, or KCNMB4) can associate with the tetrametic channel.
BK channels are a prime example of modular protein evolution. Each BK channel alpha subunit consists of (from N- to C-terminal):
- A unique transmembrane domain (S0)[7] that precedes the 6 transmembrane domains (S1-S6) conserved in all voltage-dependent K+ channels.
- A voltage sensing domain (S1-S4).
- A K+ channel pore domain (S5, selectivity filter, and S6).
- A cytoplasmic C-terminal domain (CTD) consisting of a pair of RCK (Regulator of Conductance of K+) domains that assemble into an octameric gating ring on the intracellular side of the tetrameric channel.[8][9][10][11][12] The CTD contains four primary binding sites for Ca2+, called "calcium bowls", encoded within the second RCK domain of each monomer.[2][8][12][13]
Available X-ray structures:
- 3MT5 - Crystal Structure of the Human BK Gating Apparatus[2]
- 3NAF - Structure of the Intracellular Gating Ring from the Human High-conductance Ca2+ gated K+ Channel (BK Channel)[8]
- 3U6N - Open Structure of the BK channel Gating Ring[13]
Pharmacology
BK channels are pharmacological targets for the treatment of several medical disorders including stroke[14] and overactive bladder.[15] Although pharmaceutical companies have attempted to develop synthetic molecules targeting BK channels,[16] their efforts have proved largely ineffective. For instance, BMS-204352, a molecule developed by Bristol-Myers Squibb, failed to improve clinical outcome in stroke patients compared to placebo.[17] However, BKCa channels are reduced in patients suffering from the Fragile X syndrome[18] and the agonist, BMS-204352, corrects some of the deficits observed in Fmr1 knockout mice, a model of Fragile X syndrome.[19]
BK channels have also been found to be activated by exogenous pollutants and endogenous gasotransmitters carbon monoxide[20][21] and hydrogen sulphide.[22]
BK channels can be readily inhibited by a range of compounds including tetraethylammonium (TEA), paxilline,[23] Limbatustoxin, and iberiotoxin.[24]
The BKCa-channel blocker GAL-021 has been investigated for potential use in inhibiting opioid induced respiratory depression without affecting analgesia.[25][26]
See also
- Calcium-activated potassium channel subunit alpha-1
- Calcium-activated potassium channel
- Voltage-gated potassium channel
References
- ↑ Miller C (2000). "An overview of the potassium channel family". Genome Biology. 1 (4): REVIEWS0004. doi:10.1186/gb-2000-1-4-reviews0004. PMC 138870
 . PMID 11178249.
. PMID 11178249. - 1 2 3 Yuan P, Leonetti MD, Pico AR, Hsiung Y, MacKinnon R (Jul 2010). "Structure of the human BK channel Ca2+-activation apparatus at 3.0 A resolution". Science. 329 (5988): 182–6. doi:10.1126/science.1190414. PMC 3022345
 . PMID 20508092.
. PMID 20508092. - ↑ Kyle BD (Aug 2014). "Ion Channels of the Mammalian Urethra". Channels. 8 (5): 393–401. doi:10.4161/19336950.2014.954224. PMC 4594508
 . PMID 25483582.
. PMID 25483582. - ↑ EntrezGene 3778
- ↑ Davies AG, Pierce-Shimomura JT, Kim H, VanHoven MK, Thiele TR, Bonci A, Bargmann CI, McIntire SL (Dec 2003). "A central role of the BK potassium channel in behavioral responses to ethanol in C. elegans". Cell. 115 (6): 655–66. doi:10.1016/S0092-8674(03)00979-6. PMID 14675531.
- ↑ Alaimo JT, Davis SJ, Song SS, Burnette CR, Grotewiel M, Shelton KL, Pierce-Shimomura JT, Davies AG, Bettinger JC (Nov 2012). "Ethanol metabolism and osmolarity modify behavioral responses to ethanol in C. elegans". Alcoholism: Clinical and Experimental Research. 36 (11): 1840–50. doi:10.1111/j.1530-0277.2012.01799.x. PMC 3396773
 . PMID 22486589.
. PMID 22486589. - ↑ Wallner M, Meera P, Toro L (Dec 1996). "Determinant for beta-subunit regulation in high-conductance voltage-activated and Ca(2+)-sensitive K+ channels: an additional transmembrane region at the N terminus". Proceedings of the National Academy of Sciences of the United States of America. 93 (25): 14922–7. doi:10.1073/pnas.93.25.14922. PMC 26238
 . PMID 8962157.
. PMID 8962157. - 1 2 3 Wu Y, Yang Y, Ye S, Jiang Y (Jul 2010). "Structure of the gating ring from the human large-conductance Ca(2+)-gated K(+) channel". Nature. 466 (7304): 393–7. doi:10.1038/nature09252. PMC 2910425
 . PMID 20574420.
. PMID 20574420. - ↑ Jiang Y, Pico A, Cadene M, Chait BT, MacKinnon R (Mar 2001). "Structure of the RCK domain from the E. coli K+ channel and demonstration of its presence in the human BK channel". Neuron. 29 (3): 593–601. doi:10.1016/S0896-6273(01)00236-7. PMID 11301020.
- ↑ Pico AR (2003). RCK domain model of calcium activation in BK channels (PhD thesis). New York: The Rockfeller University.
- ↑ Yusifov T, Savalli N, Gandhi CS, Ottolia M, Olcese R (Jan 2008). "The RCK2 domain of the human BKCa channel is a calcium sensor". Proceedings of the National Academy of Sciences of the United States of America. 105 (1): 376–381. doi:10.1073/pnas.0705261105. PMC 2224220
 . PMID 18162557.
. PMID 18162557. - 1 2 Schreiber M, Salkoff L (Sep 1997). "A novel calcium-sensing domain in the BK channel". Biophysical Journal. 73 (3): 1355–63. doi:10.1016/S0006-3495(97)78168-2. PMC 1181035
 . PMID 9284303.
. PMID 9284303. - 1 2 Yuan P, Leonetti MD, Hsiung Y, MacKinnon R (Jan 2012). "Open structure of the Ca2+ gating ring in the high-conductance Ca2+-activated K+ channel". Nature. 481 (7379): 94–7. doi:10.1038/nature10670. PMC 3319005
 . PMID 22139424.
. PMID 22139424. - ↑ Gribkoff VK, Starrett JE, Dworetzky SI (Apr 2001). "Maxi-K potassium channels: form, function, and modulation of a class of endogenous regulators of intracellular calcium". The Neuroscientist. 7 (2): 166–77. doi:10.1177/107385840100700211. PMID 11496927.
- ↑ Layne JJ, Nausch B, Olesen SP, Nelson MT (Feb 2010). "BK channel activation by NS11021 decreases excitability and contractility of urinary bladder smooth muscle". American Journal of Physiology. Regulatory, Integrative and Comparative Physiology. 298 (2): R378–84. doi:10.1152/ajpregu.00458.2009. PMC 2828174
 . PMID 19923353.
. PMID 19923353. - ↑ Gribkoff VK, Winquist RJ (May 2005). "Voltage-gated cation channel modulators for the treatment of stroke". Expert Opinion on Investigational Drugs. 14 (5): 579–92. doi:10.1517/13543784.14.5.579. PMID 15926865.
- ↑ Jensen BS (2002). "BMS-204352: a potassium channel opener developed for the treatment of stroke". CNS Drug Reviews. 8 (4): 353–60. doi:10.1111/j.1527-3458.2002.tb00233.x. PMID 12481191.
- ↑ Laumonnier F, Roger S, Guérin P, Molinari F, M'rad R, Cahard D, Belhadj A, Halayem M, Persico AM, Elia M, Romano V, Holbert S, Andres C, Chaabouni H, Colleaux L, Constant J, Le Guennec JY, Briault S (2006). "Association of a functional deficit of the BKCa channel, a synaptic regulator of neuronal excitability, with autism and mental retardation". The American Journal of Psychiatry. 163 (9): 1622–1629. doi:10.1176/ajp.2006.163.9.1622. PMID 16946189.
- ↑ Hébert B; Pietropaolo S; Même S; Laudier B; Laugeray A; Doisne N; Quartier A; Lefeuvre S; Got L; Cahard D; Laumonnier F; Crusio WE; Pichon J; Menuet A; Perche O; Briault S (2014). "Rescue of fragile X syndrome phenotypes in Fmr1 KO mice by a BKCa channel opener molecule". Orphanet Journal of Rare Diseases. 9: 124. doi:10.1186/s13023-014-0124-6. PMC 4237919
 . PMID 25079250.
. PMID 25079250. - ↑ Dubuis E, Potier M, Wang R, Vandier C (Feb 2005). "Continuous inhalation of carbon monoxide attenuates hypoxic pulmonary hypertension development presumably through activation of BKCa channels". Cardiovascular Research. 65 (3): 751–61. doi:10.1016/j.cardiores.2004.11.007. PMID 15664403.
- ↑ Hou S, Xu R, Heinemann SH, Hoshi T (Mar 2008). "The RCK1 high-affinity Ca2+ sensor confers carbon monoxide sensitivity to Slo1 BK channels". Proceedings of the National Academy of Sciences of the United States of America. 105 (10): 4039–43. doi:10.1073/pnas.0800304105. PMC 2268785
 . PMID 18316727.
. PMID 18316727. - ↑ Sitdikova GF, Weiger TM, Hermann A (Feb 2010). "Hydrogen sulfide increases calcium-activated potassium (BK) channel activity of rat pituitary tumor cells". Pflügers Archiv. 459 (3): 389–97. doi:10.1007/s00424-009-0737-0. PMID 19802723.
- ↑ "Paxilline, from Fermentek". Archived from the original on 2008-05-17.
- ↑ Candia S, Garcia ML, Latorre R (Aug 1992). "Mode of action of iberiotoxin, a potent blocker of the large conductance Ca(2+)-activated K+ channel". Biophysical Journal. 63 (2): 583–90. doi:10.1016/S0006-3495(92)81630-2. PMC 1262182
 . PMID 1384740.
. PMID 1384740. - ↑ McLeod JF, Leempoels JM, Peng SX, Dax SL, Myers LJ, Golder FJ. GAL-021, a new intravenous BKCa-channel blocker, is well tolerated and stimulates ventilation in healthy volunteers. Br J Anaesth. 2014 Nov;113(5):875-83. doi: 10.1093/bja/aeu182 PMID 24989775
- ↑ Golder, Francis J.; Dax, Scott; Baby, Santhosh M.; Gruber, Ryan; Hoshi, Toshinori; Ideo, Courtney; Kennedy, Andrew; Peng, Sean; Puskovic, Veljko; Ritchie, David; Woodward, Richard; Wardle, Robert L.; Van Scott, Michael R.; Mannion, James C.; MacIntyre, D. Euan (2015). "Identification and Characterization of GAL-021 as a Novel Breathing Control Modulator". Anesthesiology. 123 (5): 1093–1104. doi:10.1097/ALN.0000000000000844. ISSN 0003-3022.
External links
- BK Channels at the US National Library of Medicine Medical Subject Headings (MeSH)
- "Calcium-Activated Potassium Channels". IUPHAR Database of Receptors and Ion Channels. International Union of Basic and Clinical Pharmacology.