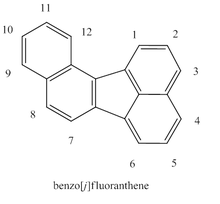
Benzo(j)fluoranthene
fluoranthene.png) | |
| Identifiers | |
|---|---|
| 205-82-3 | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 8798 |
| ECHA InfoCard | 100.005.374 |
| KEGG | C19196 |
| |
| |
| Properties | |
| C20H12 | |
| Molar mass | 252.3093 |
| Appearance | solid |
| Density | 1.286 g/cm3 |
| Melting point | 165 °C (329 °F; 438 K) |
| Hazards | |
| Flash point | 228.6 °C (443.5 °F; 501.8 K) |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Natural occurrence
Benzo[j]fluoranthene is an organic compound with the chemical formula C20H12. Classified as a polycyclic aromatic hydrocarbon (PAH), it is a colourless solid that is poorly soluble in most solvents. Impure samples can appear off white. Closely related isomeric compounds include benzo(a)fluoranthene (BaF), benzo(b)fluoranthene (BbF), benzo(e)fluoranthene (BeF), and benzo(k)fluoranthene (BkF). BjF is present in fossil fuels and is released during incomplete combustion of organic matter. It has been traced in the smoke of cigarettes, exhaust from gasoline engines, emissions from the combustion of various types of coal and emissions from oil heating,[1] as well as an impurity in some oils such as soybean oil.[2] More than 20% of the carbon in the universe may be associated with PAHs, including the benzofluoranthenes.[3]
Structure and synthesis
BjF consists of two naphthalene-like structures which are fused by a cyclopentane structure. This cyclopentane is not included in the aromaticity of the molecule. BjF can be obtained when either 2-(1-chloroethenyl)benzo[c]phenanthrene or 6-(1-chloroethenyl)chrysene is treated by flash vacuum thermolysis (FVT) at high temperatures (>900 °C) followed by ring rearrangements (ring contraction/expansion) to selectively yield BjF.[4] benzo[k]fluoranthene may also be converted via similar processes to BjF by FVT at temperatures ≥ 1100 °C (6% yield) or ≥ 1200 °C (11% yield) with 38% mass recovery.[5]
Reactivity
BjF can be functionalized by means of electrophilic aromatic substitution. In the body it is metabolized into phenols (3,4,6 or 10 hydroxy), dihydrodiols (4,5 and 9,10) and 4,5-dione[6](fig. 1).
Mechanism of action
BjF is categorized by the IARC as possibly carcinogenic to human beings, like many other PAHs, on the basis of sufficient evidence in animals.[7] For example BjF is active as a tumor initiator on mouse skin and is carcinogenic in both mouse skin and in rat lungs. Recently, BjF was also found to induce tumors in newborn mouse lung and liver.[8] The mechanism of actions of BjF is similar to other PAHs. The diolepoxide mechanism involves formation of stable and unstable DNA adducts, mainly at G and A, which can lead to mutations in proto-oncogenes (RAS) and tumour-suppressor genes (P53). Many polycyclic aromatic hydrocarbon diolepoxides and their precursor diols and epoxides are tumorigenic in animals. The radical cation mechanism involves generation of unstable adducts at G and A, leading to apurinic sites and mutations in HRAS. Orthoquinone formation could lead to stable and unstable DNA adducts and generation of reactive oxygen species, inducing mutations in P53.[9]
Metabolism
Despite the ubiquitous occurrence of benzofluoranthenes in the environment, little is known about their metabolic activation. Only one such study has appeared in which the binding of dibenzo(a,e)fluoranthene to nucleic acids was investigated. Here the potential of BjF-9,10-dihydrodiol" and BkF-8,9-dihydrodiol to contribute to the mutagenicity of BjF and BkF toward S. typhimurium TA 100 has been evaluated.[10]
A high-pressure liquid chromatogram of the metabolites of BjF formed in vitro is shown in figure 2. Six bands of metabolites were collected and tested for mutagenicity toward S. typhimurium TA 100 with activation. The results are summarized in Table 1. Band 3 had the highest mutagenic activity, suggesting that this band contained proximate mutagenic metabolites of benzo(y)fluoranthene (ByF). When tested separately, each of Peaks A and B of Band 3 was mutagenic (Table 1). Dihydrodiols are proximate mutagenic metabolites of BjF. BjF-9,10-dihydrodiol was mutagenic only in the presence of rat liver homogenate, indicating that its mutagenic activities resulted from conversion to dihydrodiol-epoxides, as observed with other hydrocarbons. Other metabolites may also contribute to the mutagenicity of BjF. This includes BjF-9,10-epoxide which is the likely precursor to the observed dihydrodiols, but it was not isolated or synthesized. The structure of BjF-9,10-dihydrodiol is similar to those of dihydrodiols which are proximate forms of other carcinogenic polycyclic aromatic hydrocarbons and can form dihydrodiol- epoxides with one carbon of the epoxide ring in a 3-sided "bay region". The presumed ultimate mutagen derived from B(j)F9,10-dihydrodiol, 9,10-dihydro-9,10-dihydroxy- 11,12-epoxybenzo(j)-fluoranthene would have the epoxide ring in a 4-sided, "pseudo-bay region," in which the additional side is derived from the 5-membered ring. Such a dihydrodiol-epoxide would not be expected to be a potent carcinogen. B(j)F-9,10-dihydrodiol is currently being bioassayed on mouse skin for carcinogenicity. It is the conversion of trans-4,5-dihydro-4,5-dihydroxybenzo[j]fluoranthene to anti-4,5-dihydroxy-5,6a-epoxy-4,5,6,6a- tetrahydrobenzo[j]fluoranthene that is principally associated with DNA adduct formation in mouse skin.[11][12]
Toxicity
PAHs
One of the earliest connection between PAH’s, combustion, and cancer was established by Cook and co-workers with the isolation of the carcinogen benzo[a]pyrene from coal tar extract.[13] Benzo[a]pyrene now has been well characterized in toxicology reports and is a known potent carcinogen.[14] Benzo[a]pyrene requires metabolic activation to become, ultimately, BPDE ( (±)-anti-7β,8α-dihydroxy-9α,10α-epoxy-7,8,9,10-tetrahydrobenzo[a]pyrene ) which binds to the DNA to form a covalent trans adducts at the N2 position of guanine.[15] Hereafter binding to DNA at cancer hotspots, especially in the P53 tumour suppressor gene at codons: 157, 248 and 273 (figure 3), it has the possibility of inducing lung cancer.[16] Structural similarity of PAHs contributes to the similarity in metabolism, biotransformation and toxicology. Benzo[a]pyrene has been extensively reviewed and is used as a model for the toxicology and metabolism of other PAHs.[17]
Benzo[j]fluoranthene
Specific studies on BjF showed that it exhibits mutagenic toxicity in S. typhimurium TA98 and TA1000 under the presence of microsomal activation.[18] BjF can form DNA-adducts, covalently binding of chemicals to DNA can result in strand breaks and DNA damage, which ultimately leads to mutations.[19] In mice studies BjF induced tumorigenic activity on the skin, lung adenomas and liver adenomas/hepatomas.[20][21][22][23] Lung implantation of BjF also induced lung epidermoid carcinomas in 3-month-old female rats.[24] Tail vein injection of BjF also causes covalently binding to mouse hemoglobin and serum proteins, with binding to serum proteins being 10-fold higher than to hemoglobin.[25]
References
- ↑ Author unknown (23 June 2005) “Benzo(j)fluoranthene”. TOXNET http://toxnet.nlm.nih.gov/cgi-bin/sis/search2/f?./temp/~ZHcJmU:1 (last consulted on: 19 March 2015)
- ↑ Author unknown (date unknown) “Benzo[j]fluoranthene” Sigma-Aldrich http://www.sigmaaldrich.com/catalog/product/Fluka/BCR049?lang=en®ion=NL[] (last consulted on: 19 March 2015)
- ↑ R. Hoover (February 21, 2014). "Need to Track Organic Nano-Particles Across the Universe? NASA's Got an App for That". NASA. Retrieved February 22, 2014.
- ↑ M. Sarobe et al. (1 January 1997) “High temperature gas phase syntheses of C20H12 cyclopenta-fused polycyclic aromatic hydrocarbons: benz[l]acephenanthrylene and benz[j]acephenanthrylene and their selective rearrangement to benzo[j]fluoranthene” Journal of the Chemical Society, Perkin Trans. 2
- ↑ M. Sarobe et al. (1999) “Flash Vacuum Thermolysis of Acenaphtho[1,2-α]acenaphthylene, Fluoranthene, Benzo[k]- and Benzo[j]fluoranthene 2 Homolytic Scission of Carbon2Carbon Single Bonds of Internally Fused Cyclopenta Moieties at T ≥ 1100°C” European Journal of Organic Chemistry
- ↑ E.H. Weyand et al. (1993) “Detection of the Major DNA Adducts of Benzo[j]fluoranthene in Mouse Skin: Nonclassical Dihydrodiol Epoxides” Chemical Research in Toxicology 6
- ↑ K.Straif et al. (December 2005) “Carcinogenicity of polycyclic aromatic hydrocarbons” THE LANCET Oncology, vol.6, issue 12
- ↑ J.E. Rice et al. (1st of December 1987) “Identification of Tumorigenic Metabolites of Benzo[/]fluoranthene Formed in Vivo in Mouse Skin” Cancer Research 47, 6166-6170
- ↑ K.Straif et al. (December 2005) “Carcinogenicity of polycyclic aromatic hydrocarbons” The Lancet Oncology, vol.6, issue 12
- ↑ E.J. Lavoie et al. (December 1980) “Identification of Mutagenic Dihydrodiols as Metabolites of Benzo(j)fluoranthene and Benzo(k)fluoranthene” Cancer Research 40, 4528-4532
- ↑ E.H. Weyand et al. (1993) “Detection of the Major DNA Adducts of Benzo[j]fluoranthene in Mouse Skin: Nonclassical Dihydrodiol Epoxides” Chemical Research in Toxicology 6
- ↑ E.J. LaVoie et al. (1993) “Exceptional tumor-initiating activity of 4-fluorobenzoljlfluoroanthene on mouse skin: comparison with benzo[j]fluoranthene, l0-fluoro-benzoIj]fluoranthene, benzo[a]pyrene, dibenzo[a,l]pyrene and 7,12-dimethylbenz[a]anthracene” Cancer Letters 70:7-14.
- ↑ Cook, James Wilfred, C. L. Hewett, and I. Hieger. "106. The isolation of a cancer-producing hydrocarbon from coal tar. Parts I, II, and III." Journal of the Chemical Society (1933): 395-405.
- ↑ Denissenko, Mikhail F., et al. "Preferential formation of benzo [a] pyrene adducts at lung cancer mutational hotspots in P53." Science 274.5286 (1996): 430-432.
- ↑ Singer, B., and D. Grunberger. "Metabolic activation of carcinogens and mutagens." Molecular Biology of Mutagens and Carcinogens. Springer US, 1983. 97-141. ISBN 978-1-4613-3772-0
- ↑ Denissenko, Mikhail F., et al. "Preferential formation of benzo [a] pyrene adducts at lung cancer mutational hotspots in P53." Science 274.5286 (1996): 430-432.
- ↑ Agency for Toxic Substances and Disease Registry (ATSDR). (1995) “Toxicological profile for Polycyclic Aromatic Hydrocarbons (PAHs)” Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service.
- ↑ E.J. Lavoie et al. (December 1980) “Identification of Mutagenic Dihydrodiols as Metabolites of Benzo(j)fluoranthene and Benzo(k)fluoranthene” Cancer Research 40, 4528-4532
- ↑ Agency for Toxic Substances and Disease Registry (ATSDR). (1995) “Toxicological profile for Polycyclic Aromatic Hydrocarbons (PAHs)” Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service.
- ↑ LaVoie, Edmond J., et al. "Tumour initiating activity of dihydrodiols of benzo [b] fluoranthene, benzo [j] fluoranthene, and benzo [k] fluoranthene." Carcinogenesis 3.1 (1982): 49-52.
- ↑ Habs, M., D. Schmähl, and J. Misfeld. "Local carcinogenicity of some environmentally relevant polycyclic aromatic hydrocarbons after lifelong topical application to mouse skin." Archiv für Geschwulstforschung 50.3 (1979): 266-274.
- ↑ Wynder, Ernest L., and Dietrich Hoffmann. "The carcinogenicity of benzofluoranthenes." Cancer 12.6 (1959): 1194-1199.
- ↑ Weyand, E. H., et al. "Effect of fluorine substitution on benzo [j] fluoranthene genotoxicity." Chemico-Biological Interactions 84.1 (1992): 37-53
- ↑ Deutsch-Wenzel, Reintraud P., et al. "Experimental studies in rat lungs on the carcinogenicity and dose-response relationships of eight frequently occurring environmental polycyclic aromatic hydrocarbons." Journal of the National Cancer Institute 71.3 (1983): 539-544
- ↑ Agency for Toxic Substances and Disease Registry (ATSDR). (1995) “Toxicological profile for Polycyclic Aromatic Hydrocarbons (PAHs)” Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service.