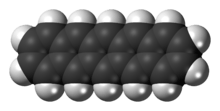
Pentacene
| | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Pentacene | |
| Identifiers | |
| 135-48-8 | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:33148 |
| ChemSpider | 8347 |
| ECHA InfoCard | 100.004.722 |
| PubChem | 8671 |
| |
| |
| Properties | |
| C22H14 | |
| Molar mass | 278.36 g/mol |
| Appearance | Dark blue powder |
| Density | 1.3 g/cm3 |
| Melting point | > 300 °C; sublimes at 372 °C |
| Structure | |
| Triclinic | |
| P-1 | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Pentacene is a polycyclic aromatic hydrocarbon consisting of five linearly-fused benzene rings. This highly conjugated compound is an organic semiconductor. The compound generates excitons upon absorption of ultra-violet (UV) or visible light; this makes it very sensitive to oxidation. For this reason, this compound, which is a purple powder, slowly degrades upon exposure to air and light.
Structurally, pentacene is one of the linear acenes, the previous one being tetracene (four fused benzene rings) and the next one being hexacene (six fused benzene rings). In August 2009, a group of researchers from IBM published experimental results of imaging a single molecule of pentacene using an atomic force microscope.[1][2] In July 2011, they used a modification of scanning tunneling microscopy to experimentally determine the shapes of the highest occupied and lowest unoccupied molecular orbitals.[3][4]
In 2012, pentacene-doped p-terphenyl was shown to be effective as the amplifier medium for a room-temperature maser.[5]
In February 2014, NASA announced a greatly upgraded database for tracking polycyclic aromatic hydrocarbons (PAHs), including Pentacene, in the universe. According to scientists, more than 20% of the carbon in the universe may be associated with PAHs, possible starting materials for the formation of life. PAHs seem to have been formed shortly after the Big Bang, are widespread throughout the universe, and are associated with new stars and exoplanets.[6]
Synthesis
_STM.jpg)

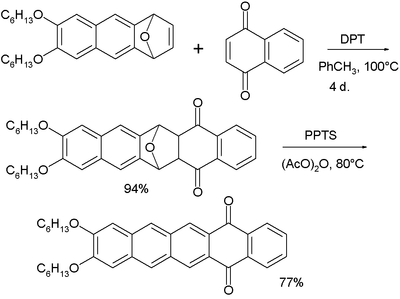
A classic method for pentacene synthesis is by the Elbs reaction. Pentacenes can also be prepared in the laboratory by extrusion of a small volatile component. In one such experimental procedure carbon monoxide is liberated from a precursor at 150 °C.[8] The precursor is reported to have some solubility in chloroform and is therefore amenable to spin coating. Pentacene is soluble in hot chlorinated benzenes, such as 1,2,4-trichlorobenzene, from which it can be recrystallized to form platelets.
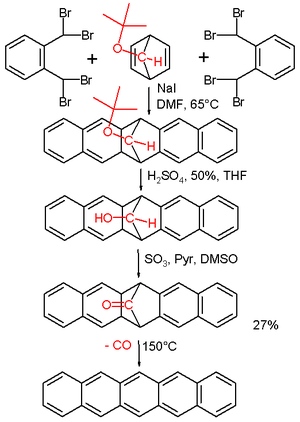 Pentacene synthesis
Pentacene synthesis
Pentacene derivatives
Monomeric pentacene derivatives
6,13-Substituted pentacenes are accessible through pentacenequinone by reaction with an aryl or alkynyl nucleophile (for example Grignard or organolithium reagents) followed by reductive aromatization.[9][10][11] Another method is based on homologization of diynes by transition metals (through zirconacyclopentadienes) [12][13][14][15][16] Functionalization of pentacene has allowed for control of the solid-state packing of this chromophore.[17][18] The choice of the substituents (both size and location of substitution on the pentacene) influences the solid-state packing and can be used to control whether the compound adopts 1-dimensional or 2-dimensional cofacial pi-stacking in the solid-state, as opposed to the herringbone packing observed for pentacene.
Although pentacene's structure resembles that of other aromatic compounds like anthracene, its aromatic properties are poorly defined; as such, pentacene and its derivatives are the subject of much research.
A tautomeric chemical equilibrium exists between 6-methylene-6,13-dihydropentacene and 6-methylpentacene.
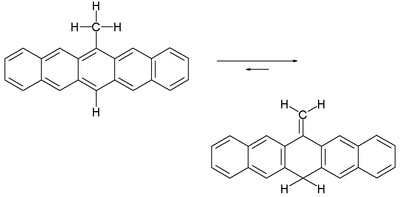 6-methylpentacene equilibrium
6-methylpentacene equilibrium
This equilibrium is entirely in favor of the methylene compound. Only by heating a solution of the compound to 200 °C does a small amount of the pentacene develop, as evidenced by the emergence of a red-violet color. According to one study[19] the reaction mechanism for this equilibrium is not based on an intramolecular 1,5-hydride shift, but on a bimolecular free radical hydrogen migration. In contrast, isotoluenes with the same central chemical motif easily aromatize.
Pentacene reacts with elemental sulfur in 1,2,4-trichlorobenzene to the compound hexathiapentacene.[20] X-ray crystallography shows that all the carbon-to-sulfur bond lengths are roughly equal (170 pm); from this, it follows that resonance structures B and C with complete charge separation are more significant than structure A.
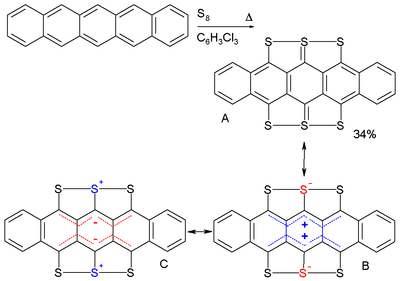 Hexathiapentacene
Hexathiapentacene
In the crystal phase the molecules display aromatic stacking interactions, whereby the distance between some sulfur atoms on neighboring molecules can become less (337 pm) than the sum of two Van der Waals radii (180 pm)
Like the related tetrathiafulvalene, this compound is studied in the field of organic semiconductors.
The acenes may appear as planar and rigid molecules, but in fact they can be very distorted. The pentacene depicted below:[21]
 Twisted acenes
Twisted acenes
has an end-to end twist of 144° and is sterically stabilized by the six phenyl groups. The compound can be resolved into its two enantiomers with an unusually high reported optical rotation of 7400° although racemization takes place with a chemical half-life of 9 hours.
Oligomers and polymers of pentacene
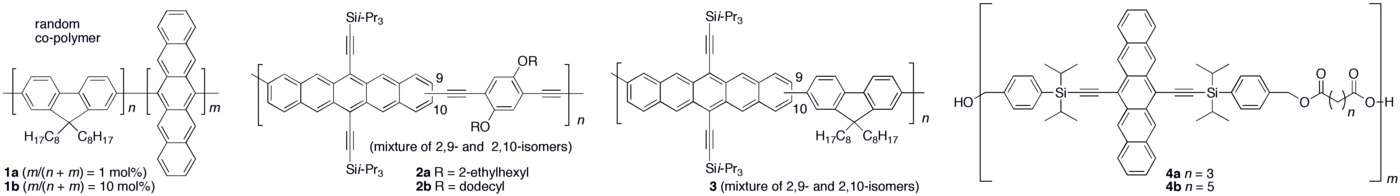
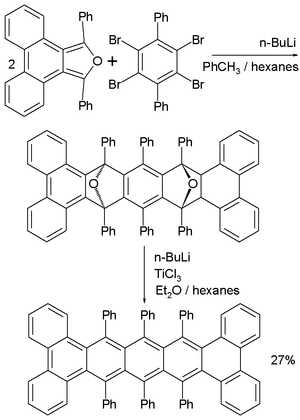
Oligomers and polymers based on pentacene have been explored both synthetically as well as in device application settings.[22][23] Polymer light emitting diodes (PLEDs) have been constructed using conjugated copolymers (1a–b) containing fluorene and pentacene.[24] A few other conjugated pentacene polymers (2a–b and 3) have been realized based on Sonogashira and Suzuki coupling reactions of a dibromopentacene monomer.[25][26] Non-conjugated pentacene-based polymers have been synthesized via esterification of a pentacene diol monomer with bis-acid chlorides to from polymers 4a–b.[27][28]
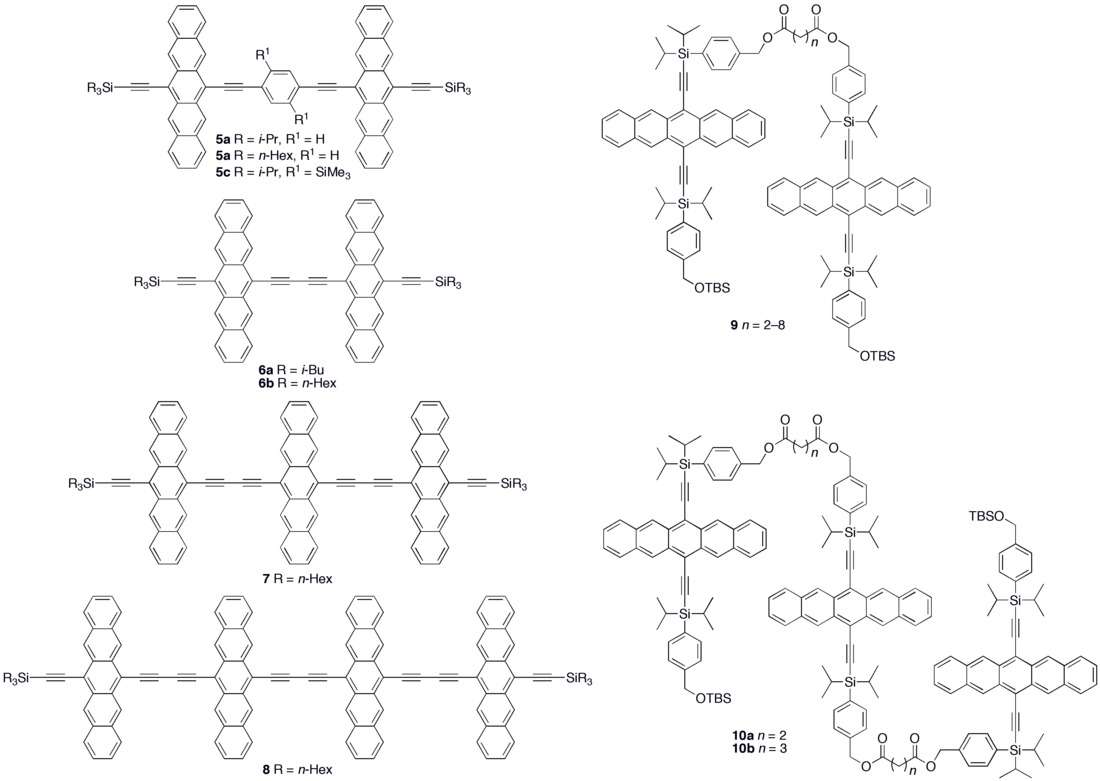
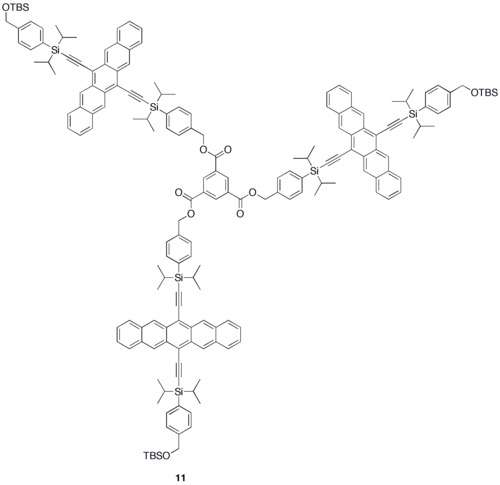
Various synthetic strategies have been employed to form conjugated oligomers of pentacene 5a–c including a one-pot-four-bond forming procedure which provided a solution-processable conjugated pentacene dimer (5c) which exhibited photoconductive gain >10,[29] placing its performance within the same order of magnitude as thermally evaporated films of non-functionalized pentacene which exhibited photoconductive gain >16 using analogous measurement techniques.[30] A modular synthetic method to conjugated pentacene di-, tri- and tetramers (6–8) has been reported which is based on homo- and cross-coupling reactions of robust dehydropentacene intermediates.[31] Non-conjugated oligomers 9–10 based on pentacene have been synthesized,[27][28] including dendrimers 9–10 with up to 9 pentacene moieties per molecule with molar absorptivity for the most intense absorption > 2,000,000 M−1•cm−1. Dendrimers 11–12 were shown to have improved performance in devices compared to analogous pentacene-based polymers 4a–b in the context of photodetectors.[32]
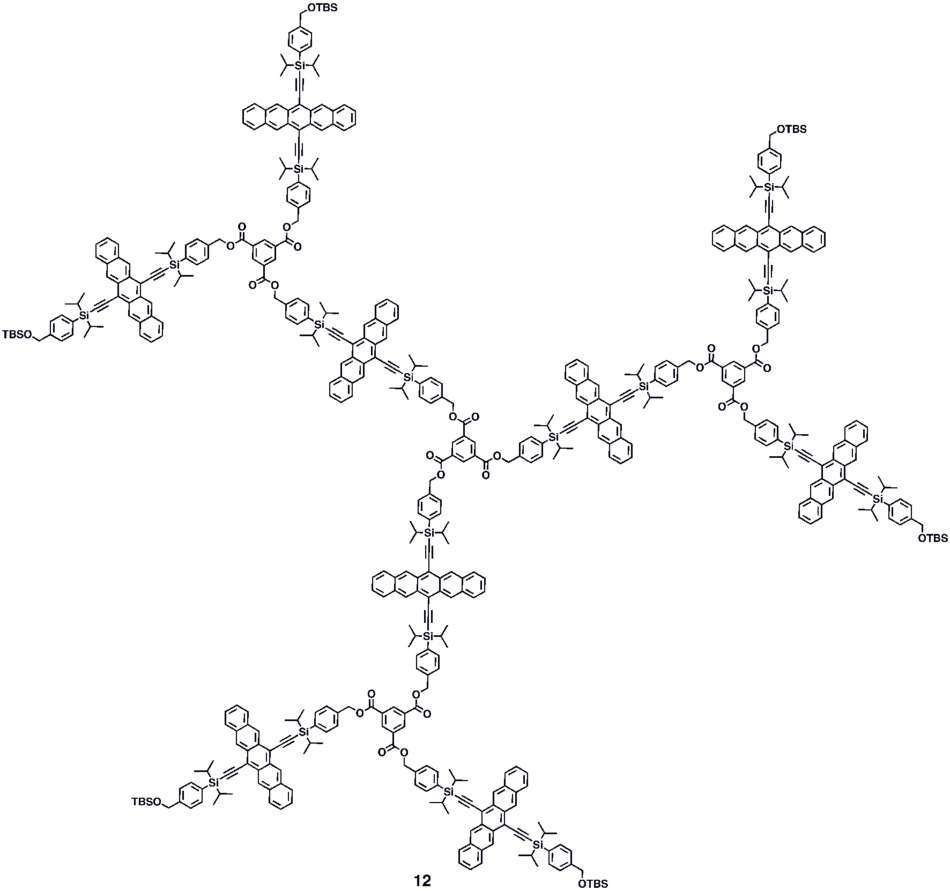
Materials research
Pentacenes have been examined as potential dichroic dyes. The pentacenoquinone displayed below is fluorescent and when mixed with liquid crystal E7 mixture a dichroic ratio of 8 is reached.[33][34] Longer acenes align better in the nematic liquid crystal phase.
 Fluorescent acenequinones
Fluorescent acenequinones
Combined with buckminsterfullerene, pentacene is used in the development of organic photovoltaic prototypes.[35][36]
Pentacene is a popular choice for research on organic thin-film transistors and OFETs, being one of the most thoroughly investigated conjugated organic molecules with a high application potential due to a hole mobility in OFETs of up to 5.5 cm2/(V·s), which exceeds that of amorphous silicon.[37][38][39]
Pentacene, as well as other organic conductors, is subject to rapid oxidation in air, which precludes commercialization. If the pentacene is preoxidized, the pentacene-quinone is a potential gate insulator, then the mobility can approach that of rubrene – the highest-mobility organic semiconductor – namely, 40 cm2/(V·s). This pentacene oxidation technique is akin to the silicon oxidation used in the silicon electronics.[38]
See also
References
- ↑ "Single molecule's stunning image". BBC News. 2009-08-28. Retrieved 2009-08-28.
- ↑ Gross, L.; Mohn, F; Moll, N; Liljeroth, P; Meyer, G (2009). "The Chemical Structure of a Molecule Resolved by Atomic Force Microscopy". Science. 325 (5944): 1110–4. Bibcode:2009Sci...325.1110G. doi:10.1126/science.1176210. PMID 19713523.
- ↑ Krieger, K. (23 August 2011). "Molecules Imaged Most Intimately". Science Now.
- ↑ Gross, L.; Moll, N.; Mohn, F.; Curioni, A.; Meyer, G.; Hanke, F.; Persson, M. (2011). "High-Resolution Molecular Orbital Imaging Using a p-Wave STM Tip". Phys. Rev. Lett. 107 (8): 86101–86104. Bibcode:2011PhRvL.107h6101G. doi:10.1103/PhysRevLett.107.086101.
- ↑ Brumfiel, G. (2012). "Microwave laser fulfills 60 years of promise". Nature. doi:10.1038/nature.2012.11199.
- ↑ Hoover, Rachel (February 21, 2014). "Need to Track Organic Nano-Particles Across the Universe? NASA's Got an App for That". NASA. Retrieved February 22, 2014.
- ↑ Dinca, L. E.; De Marchi, F.; MacLeod, J. M.; Lipton-Duffin, J.; Gatti, R.; Ma, D.; Perepichka, D. F.; Rosei, F. (2015). "Pentacene on Ni(111): Room-temperature molecular packing and temperature-activated conversion to graphene". Nanoscale. 7 (7): 3263–9. doi:10.1039/C4NR07057G. PMID 25619890.
- ↑ Chen, Kew-Yu; Hsieh, HH; Wu, CC; Hwang, JJ; Chow, TJ (2007). "A new type of soluble pentacene precursor for organic thin-film transistors". Chemical Communications (10): 1065–7. doi:10.1039/b616511g. PMID 17325807.
- ↑ Allen, C. F. H.; Bell, Alan (1942). "Action of Grignard Reagents on Certain Pentacenequinones, 6,13-Diphenylpentacene". Journal of the American Chemical Society. 64 (6): 1253–1260. doi:10.1021/ja01258a005.
- ↑ Maulding, D. R.; Roberts, Bernard G. (1969). "Electronic absorption and fluorescence of phenylethynyl-substituted acenes". The Journal of Organic Chemistry. 34 (6): 1734–1736. doi:10.1021/jo01258a045.
- ↑ Li, Shi; Zhou, Lishan; Nakajima, Kiyohiko; Kanno, Ken-Ichiro; Takahashi, Tamotsu (2010). "Synthesis of 1,2,3,4,8,9,10,11-Octasubstituted Pentacenequinone Derivatives and their Conversion into Substituted Pentacenes". Chemistry: An Asian Journal. 5 (7): 1620–6. doi:10.1002/asia.200900754. PMID 20455241.
- ↑ Takahashi, Tamotsu; Kitamura, Masanori; Shen, Baojian; Nakajima, Kiyohiko (2000). "Straightforward Method for Synthesis of Highly Alkyl-Substituted Naphthacene and Pentacene Derivatives by Homologation". Journal of the American Chemical Society. 122 (51): 12876–12877. doi:10.1021/ja003130g.
- ↑ Takahashi, Tamotsu; Li, Shi; Huang, Wenying; Kong, Fanzhi; Nakajima, Kiyohiko; Shen, Baojian; Ohe, Takahiro; Kanno, Ken-Ichiro (2006). "Homologation Method for Preparation of Substituted Pentacenes and Naphthacenes". The Journal of Organic Chemistry. 71 (21): 7967–77. doi:10.1021/jo060923y. PMID 17025283.
- ↑ Takahashi, Tamotsu; Li, Yanzhong; Hu, Jinghan; Kong, Fanzhi; Nakajima, Kiyohiko; Zhou, Lishan; Kanno, Ken-Ichiro (2007). "Cu(I)-mediated cycloaddition reaction of zirconacyclopentadienes with fumaronitrile and application for synthesis of monocyano-substituted pentacenes". Tetrahedron Letters. 48 (38): 6726–6730. doi:10.1016/j.tetlet.2007.07.075.
- ↑ Stone, Matthew T.; Anderson, Harry L. (2007). "Three-Step Synthesis of End-Substituted Pentacenes". The Journal of Organic Chemistry. 72 (25): 9776–8. doi:10.1021/jo7017284. PMID 17999529.
- ↑ Li, Shi; Li, Zhiping; Nakajima, Kiyohiko; Kanno, Ken-Ichiro; Takahashi, Tamotsu (2009). "Double Homologation Method for Substituted Soluble Pentacenes and Dimerization Behaviours of Pentacenes". Chemistry: An Asian Journal. 4 (2): 294–301. doi:10.1002/asia.200800312. PMID 19072938.
- ↑ Anthony, J. E.; Brooks, J. S.; Eaton, D. L.; Parkin, S. R. (2001). "Functionalized Pentacene: Improved Electronic Properties from Control of Solid-State Order". Journal of the American Chemical Society. 123 (38): 9482–9483. doi:10.1021/ja0162459. PMID 11562247.
- ↑ Anthony, J. E.; Eaton, D. L.; Parkin, S. R. (2002). "A Road Map to Stable, Soluble, Easily Crystallized Pentacene Derivatives". Organic Letters. 4 (1): 15–18. doi:10.1021/ol0167356. PMID 11772079.
- ↑ Norton, Joseph E.; Northrop, BH; Nuckolls, C; Houk, KN (2006). "Why 6-Methylpentacene Deconjugates but Avoids the Thermally Allowed Unimolecular Mechanism". Organic Letters. 8 (21): 4915–8. doi:10.1021/ol062012g. PMID 17020335.
- ↑ Briseno, Alejandro L.; Miao, Q; Ling, MM; Reese, C; Meng, H; Bao, Z; Wudl, F (2006). "Hexathiapentacene: Structure, Molecular Packing, and Thin-Film Transistors". Journal of the American Chemical Society. 128 (49): 15576–7. doi:10.1021/ja066088j. PMID 17147352..
- ↑ Lu, Jun; Ho, DM; Vogelaar, NJ; Kraml, CM; Bernhard, S; Byrne, N; Kim, LR; Pascal Jr, RA (2006). "Synthesis, Structure, and Resolution of Exceptionally Twisted Pentacenes". Journal of the American Chemical Society. 128 (51): 17043–50. doi:10.1021/ja065935f. PMID 17177456.
- ↑ Lehnherr, D.; Tykwinski, R.R. (2010). "Oligomers and Polymers Based on Pentacene Building Blocks". Materials. 3 (4): 2772–2800. Bibcode:2010Mate....3.2772L. doi:10.3390/ma3042772.
- ↑ Lehnherr, D.; Tykwinski, R. R. (2011). "Conjugated Oligomers and Polymers Based on Anthracene, Tetracene, Pentacene, Naphthodithiophene, and Anthradithiophene Building Blocks". Australian Journal of Chemistry. 64 (7): 919–929. doi:10.1071/CH11169.
- ↑ Tokito, S.; Weinfurtner, K.-H.; Fujikawa, H.; Tsutsui, T.; Taga, Y. (2001). "Acene containing polyfluorenes for red, green and blue emission in organic light-emitting diodes". Proc. SPIE–Int. Opt. Soc. Eng. Organic Light-Emitting Materials and Devices IV. 4105: 69–74. doi:10.1117/12.416877.
- ↑ Okamoto, T.; Bao, Z. (2007). "Synthesis of solution-soluble pentacene-containing conjugated copolymers". Journal of the American Chemical Society. 129 (34): 10308–10309. doi:10.1021/ja0725403. PMID 17685520.
- ↑ Okamoto, T.; Okamoto, T.; Jiang, Y.; Qu, F.; Mayer, A.C.; Parmer, J.E.; McGehee, M.D.; Bao, Z. (2008). "Synthesis and characterization of pentacene– and anthradithiophene–fluorene conjugated copolymers synthesized by Suzuki reactions". Macromolecules. 41 (19): 6977–6980. Bibcode:2008MaMol..41.6977O. doi:10.1021/ma800931a.
- 1 2 Lehnherr, D.; Tykwinski, R. R. (2007). "Pentacene Oligomers and Polymers: Functionalization of Pentacene to Afford Mono-, Di-, Tri-, and Polymeric Materials". Organic Letters. 9 (22): 4583–4586. doi:10.1021/ol702094d. PMID 17918951.
- 1 2 Lehnherr, Dan; McDonald, Robert; Ferguson, Michael J.; Tykwinski, Rik R. (2008). "Synthesis of soluble oligo- and polymeric pentacene-based materials". Tetrahedron. 64 (50): 11449–11461. doi:10.1016/j.tet.2008.09.041. ISSN 0040-4020.
- ↑ Lehnherr, D.; Gao, J.; Hegmann, F. A.; Tykwinski, R. R. (2008). "Synthesis and Electronic Properties of Conjugated Pentacene Dimers". Organic Letters. 10 (21): 4779–4782. doi:10.1021/ol801886h. PMID 18823120.
- ↑ Gao, J.; Hegmann, F. A (2008). "Bulk photoconductive gain in pentacene thin films". Applied Physics Letters. 93 (22): 223306. Bibcode:2008ApPhL..93v3306G. doi:10.1063/1.3043431.
- ↑ Lehnherr, D.; Murray, A. H.; McDonald, R.; Tykwinski, R.R. (2010). "A Modular Synthetic Approach to Conjugated Pentacene Di-, Tri-, and Tetramers". Angewandte Chemie International Edition. 49 (35): 6190–6194. doi:10.1002/anie.201000555.
- ↑ Lehnherr, D.; Gao, J.; Hegmann, F. A.; Tykwinski, R. R. (2009). "Pentacene-based dendrimers: synthesis and thin film photoconductivity measurements of branched pentacene oligomers". Journal of Organic Chemistry. 74 (14): 5017–5024. doi:10.1021/jo9007089. PMID 19489566.
- ↑ Chen, Zhihua; Swager, TM (2007). "Synthesis and Characterization of Fluorescent Acenequinones as Dyes for Guest−Host Liquid Crystal Displays". Organic Letters. 9 (6): 997–1000. doi:10.1021/ol062999m. PMID 17298074.
- ↑ in the synthesis of this compound, the starting material is treated with 1,4-naphthoquinone and DPT. DTP converts the oxo-norbornadiene to an intermediary furan. The second step is oxidation by PPTS
- ↑ Dissanayake, D. M. Nanditha M. (2007). "Nanoimprinted large area heterojunction pentacene-C[sub 60] photovoltaic device". Applied Physics Letters. 90 (25): 253502. Bibcode:2007ApPhL..90y3502D. doi:10.1063/1.2749863.
- ↑ Efficiently Organic: Researchers Use Pentacene To Develop Next-generation Solar Power sciencedaily.com Link
- ↑ Norbert Koch (2007). "Organic Electronic Devices and Their Functional Interfaces". ChemPhysChem. 8 (10): 1438–55. doi:10.1002/cphc.200700177. PMID 17539032.
- 1 2 Tatsuo Hasegawa & Jun Takeya (2009). "Organic field-effect transistors using single crystals". Sci. Technol. Adv. Mater. 10 (2): 024314. Bibcode:2009STAdM..10b4314H. doi:10.1088/1468-6996/10/2/024314.
- ↑ Yoshiro Yamashita (2009). "Organic semiconductors for organic field-effect transistors". Sci. Technol. Adv. Mater. 10 (2): 024313. Bibcode:2009STAdM..10b4313Y. doi:10.1088/1468-6996/10/2/024313.
External links
- facts about pentacene, retrieved Apr. 17, 2006
- Organic transistor improves with age, New Scientist, 2 December 2007
- Pentacene Imaged, IBM images Pentacene, the first molecule imaged in detail 29 August 2009