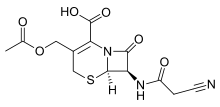
Cefacetrile
 | |
| Clinical data | |
|---|---|
| Trade names | Celospor, Celtol, Cristacef |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Intravenous, intramuscular, intramammary |
| ATC code | J01DB10 (WHO) QJ51DB10 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 23 to 38% |
| Biological half-life | 1.2 hours |
| Excretion | Renal (72%) |
| Identifiers | |
| |
| CAS Number |
10206-21-0 |
| PubChem (CID) | 91562 |
| DrugBank |
DB01414 |
| ChemSpider |
82675 |
| UNII |
FDM21QQ344 |
| KEGG |
D07629 |
| ChEMBL | CHEMBL2104099 |
| ECHA InfoCard | 100.030.449 |
| Chemical and physical data | |
| Formula | C13H13N3O6S |
| Molar mass | 339.325 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| (verify) | |
Cefacetrile (INN, also spelled cephacetrile) is a broad-spectrum first generation cephalosporin antibiotic effective in gram-positive and gram-negative bacterial infections. It is a bacteriostatic antibiotic.[1][2] Cefacetrile is marketed under the trade names Celospor, Celtol, and Cristacef,[3] and as Vetimast for the treatment of mammary infections in lactating cows.[2]
Synthesis
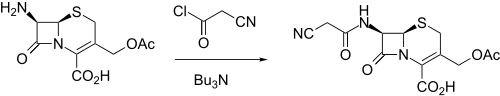
It was made by reacting 7-ACA (7-aminocephalosporanic acid) with cyanoacetyl chloride in the presence of tributylamine.
References
- ↑ "Cefacetrile Summary Report" (PDF). European Medicines Agency, Committee for Veterinary Medicinal Products. 1998.
- 1 2 Haberfeld, H, ed. (2007). Austria-Codex (in German) (2007/2008 ed.). Vienna: Österreichischer Apothekerverlag. ISBN 3-85200-183-8.
- ↑ Horiuchi, N.; Oyakawa, Y.; Oka, R.; Fujiwara, T. (1980). "Clinical evaluation of cephacetrile (Celtol) for respiratory infections (author's transl)". The Japanese journal of antibiotics. 33 (10): 1145–1155. PMID 7206219.
This article is issued from Wikipedia - version of the 8/10/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.