Estradiol
 | |
 | |
| Names | |
|---|---|
| Pronunciation | ESS-tra-DYE-ole[1][2] |
| IUPAC name
(8R,9S,13S,14S,17S)-13-Methyl-6,7,8,9,11,12,14,15,16,17-decahydrocyclopenta[a]phenanthrene-3,17-diol | |
| Other names
Estra-1,3,5(10)-triene-3,17β-diol; 17β-Estradiol | |
| Identifiers | |
| 50-28-2 | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:16469 |
| ChEMBL | ChEMBL135 |
| ChemSpider | 5554 |
| DrugBank | DB00783 |
| ECHA InfoCard | 100.000.022 |
| KEGG | D00105 |
| PubChem | 5757 |
| UNII | 4TI98Z838E |
| |
| |
| Properties | |
| C18H24O2 | |
| Molar mass | 272.38 g/mol |
| Pharmacology | |
| G03CA03 (WHO) | |
| Oral, sublingual, intranasal, topical/transdermal, vaginal, intramuscular or subcutaneous (as an ester), subdermal implant | |
| Pharmacokinetics: | |
| Oral: <5%[3] | |
| ~98%:[3][4] • Albumin: 60% • SHBG: 38% • Free: 2% | |
| Liver (via hydroxylation, sulfation, glucuronidation) | |
| Oral: 13–20 hours[3] Sublingual: 8–18 hours[5] Topical (gel): 36.5 hours[6] | |
| Urine: 54%[3] Feces: 6%[3] | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Estradiol (E2), also spelled oestradiol, is a steroid, an estrogen, and the primary female sex hormone. It is named for and is important in the regulation of the estrous and menstrual female reproductive cycles. Estradiol is essential for the development and maintenance of female reproductive tissues such as the breasts, uterus, and vagina during puberty, adulthood, and pregnancy,[7] but it also has important effects in many other tissues including bone, fat, skin, liver, and the brain. While estrogen levels in men are lower compared to women, estrogens have essential functions in men as well. Estradiol is found in most vertebrates as well as many crustaceans, insects, fish, and other animal species.[8][9]
Estradiol is produced especially within the follicles of the female ovaries, but also in other endocrine (i.e., hormone-producing) and non-endocrine tissues (e.g., including fat, liver, adrenal, breast, and neural tissues). Estradiol is biosynthesized from cholesterol through a series of chemical intermediates.[10] One principal pathway involves the generation of 4-androstenedione, which is converted into estrone by aromatase and then by 17β-hydroxysteroid dehydrogenase into estradiol. Alternatively, 4-androstenedione can be converted into testosterone, an androgen and the primary male sex hormone, which in turn can be aromatized into estradiol.
Biological function
Sexual development
The development of secondary sex characteristics in women is driven by estrogens, to be specific, estradiol. These changes are initiated at the time of puberty, most are enhanced during the reproductive years, and become less pronounced with declining estradiol support after the menopause. Thus, estradiol produces breast development, and is responsible for changes in the body shape, affecting bones, joints, and fat deposition. Fat structure and skin composition are modified by estradiol.
Female reproduction
In the female, estradiol acts as a growth hormone for tissue of the reproductive organs, supporting the lining of the vagina, the cervical glands, the endometrium, and the lining of the fallopian tubes. It enhances growth of the myometrium. Estradiol appears necessary to maintain oocytes in the ovary. During the menstrual cycle, estradiol produced by the growing follicle triggers, via a positive feedback system, the hypothalamic-pituitary events that lead to the luteinizing hormone surge, inducing ovulation. In the luteal phase, estradiol, in conjunction with progesterone, prepares the endometrium for implantation. During pregnancy, estradiol increases due to placental production. The effect of estradiol, together with estrone and estriol, in pregnancy is less clear. They may promote uterine blood flow, myometrial growth, stimulate breast growth and at term, promote cervical softening and expression of myometrial oxytocin receptors. In baboons, blocking of estrogen production leads to pregnancy loss, suggesting estradiol has a role in the maintenance of pregnancy. Research is investigating the role of estrogens in the process of initiation of labor. Actions of estradiol are required before the exposure of progesterone in the luteal phase.
Male reproduction
The effect of estradiol (and estrogens in general) upon male reproduction is complex. Estradiol is produced by action of aromatase mainly in the Leydig cells of the mammalian testis, but also by some germ cells and the Sertoli cells of immature mammals.[11] It functions (in vitro) to prevent apoptosis of male sperm cells.[12] While some studies in the early 1990s claimed a connection between globally declining sperm counts and estrogen exposure in the environment,[13] later studies found no such connection, nor evidence of a general decline in sperm counts.[14][15] Suppression of estradiol production in a subpopulation of subfertile men may improve the semen analysis.[16]
Males with certain sex chromosome genetic conditions, such as Klinefelter's syndrome, will have a higher level of estradiol.[17]
Bone
Estradiol has a profound effect on bone. Individuals without it (or other estrogens) will become tall and eunuchoid, as epiphyseal closure is delayed or may not take place. Bone structure is affected also, resulting in early osteopenia and osteoporosis.[18] Also, women past menopause experience an accelerated loss of bone mass due to a relative estrogen deficiency.[19]
Brain
Estrogens can be produced in the brain from steroid precursors. As antioxidants, they have been found to have neuroprotective function.[20]
The positive and negative feedback loops of the menstrual cycle involve ovarian estradiol as the link to the hypothalamic-pituitary system to regulate gonadotropins.[21] (See Hypothalamic–pituitary–gonadal axis.)
Estrogen is considered to play a significant role in women’s mental health, with links suggested between the hormone level, mood and well-being. Sudden drops or fluctuations in, or long periods of sustained low levels of estrogen may be correlated with significant mood-lowering. Clinical recovery from depression postpartum, perimenopause, and postmenopause was shown to be effective after levels of estrogen were stabilized and/or restored.[22][23]
Recently, the volumes of sexually dimorphic brain structures in transgender women were found to change and approximate typical female brain structures when exposed to estrogen concomitantly with androgen deprivation over a period of months,[24] suggesting that estrogen and/or androgens have a significant part to play in sex differentiation of the brain, both prenatally and later in life.
There is also evidence the programming of adult male sexual behavior in many vertebrates is largely dependent on estradiol produced during prenatal life and early infancy.[25] It is not yet known whether this process plays a significant role in human sexual behavior, although evidence from other mammals tends to indicate a connection.[26]
Gynecological cancers
Estradiol has been tied to the development and progression of cancers such as breast cancer, ovarian cancer and endometrial cancer. Estradiol affects target tissues mainly by interacting with two nuclear receptors called estrogen receptor α (ERα) and estrogen receptor β (ERβ).[27][28] One of the functions of these estrogen receptors is the modulation of gene expression. Once estradiol binds to the ERs, the receptor complexes then bind to specific DNA sequences, possibly causing damage to the DNA and an increase in cell division and DNA replication. Eukaryotic cells respond to damaged DNA by stimulating or impairing G1, S, or G2 phases of the cell cycle to initiate DNA repair. As a result, cellular transformation and cancer cell proliferation occurs.[29]
Other effects
Estradiol has complex effects on the liver. It affects the production of multiple proteins, including lipoproteins, binding proteins, and proteins responsible for blood clotting. In high amounts, it can lead to cholestasis.
Estrogen affects certain blood vessels. Improvement in arterial blood flow has been demonstrated in coronary arteries.[30]
Several benign gynecologic conditions are dependent on estrogen, such as endometriosis, leiomyomata uteri, and uterine bleeding.
Biological activity
Estradiol acts primarily as an agonist of the estrogen receptor (ER), a nuclear steroid hormone receptor. There are two subtypes of the ER, ERα and ERβ, and estradiol potently binds to and activates both of these receptors. The result of ER activation is a modulation of gene transcription and expression in ER-expressing cells, which is the predominant mechanism by which estradiol mediates its biological effects in the body. Estradiol also acts as an agonist of membrane estrogen receptors (mERs), such as GPER (GPR30), a recently discovered non-nuclear receptor for estradiol, via which it can mediate a variety of rapid, non-genomic effects.[31] Unlike the case of the ER, GPER appears to be selective for estradiol, and shows very low affinities for other endogenous estrogens, such as estrone and estriol.[32] Additional mERs besides GPER include ER-X, ERx, and Gq-mER.[33][34]
In the E2 classical pathway or estrogen classical pathway, estradiol enters the cytoplasm, where it causes dissociation of heat-shock protein (HSP). Estradiol then binds to HSP and can homodimerise (form structures of two HSP and two estradiol molecules) and then bind to specific domains on the nucleus (estrogen response element, ERE), allowing for gene transcription which can take place over hours and days.
Estradiol is about 10 times as potent as estrone and about 80 times as potent as estriol in its estrogenic effect.
Biochemistry
Biosynthesis
Estradiol, like other steroids, is derived from cholesterol. After side chain cleavage and using the Δ5 or the Δ4- pathway, Δ4-androstenedione is the key intermediary. A portion of the Δ4-androstenedione is converted to testosterone, which in turn undergoes conversion to estradiol by aromatase. In an alternative pathway, Δ4-androstenedione is aromatized to estrone, which is subsequently converted to estradiol.[36]
During the reproductive years, most estradiol in women is produced by the granulosa cells of the ovaries by the aromatization of Δ4-androstenedione (produced in the theca folliculi cells) to estrone, followed by conversion of estrone to estradiol by 17β-hydroxysteroid dehydrogenase. Smaller amounts of estradiol are also produced by the adrenal cortex, and, in men, by the testes.
Estradiol is not produced in the gonads only, in particular, fat cells produce active precursors to estradiol, and will continue to do so even after menopause.[37] Estradiol is also produced in the brain and in arterial walls.
The biosynthesis of estradiol has been observed in various other species, as indicated above, but also in such species as Phaseolus vulgaris.[38] More often referred to as "beans", consumption may equate to unintentional ingestion of estradiol. In light of this, consumption can be counterproductive to patients undergoing treatment for breast cancer, which usually includes depriving the cancer cells of estrogens. Soybeans are another bean that contains chemicals that act similarly to estrogen in the human body and also cause such interactions.
Distribution
In plasma, estradiol is largely bound to SHBG, and also to albumin. Only a fraction of 2.21% (± 0.04%) is free and biologically active, the percentage remaining constant throughout the menstrual cycle.[39]
Metabolism
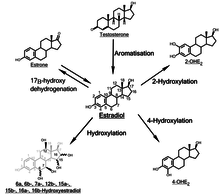
Inactivation of estradiol includes conversion to less-active estrogens, such as estrone and estriol. Estriol is the major urinary metabolite. Estradiol is conjugated in the liver by sulfate and glucuronide formation and, as such, excreted via the kidneys. Some of the water-soluble conjugates are excreted via the bile duct, and partly reabsorbed after hydrolysis from the intestinal tract. This enterohepatic circulation contributes to maintaining estradiol levels.
In the liver, estradiol is non-specifically metabolized by CYP1A2, CYP3A4, and CYP2C9 via 2-hydroxylation into 2-hydroxyestradiol, and by CYP2C9, CYP2C19, and CYP2C8 via 17β-hydroxy dehydrogenation into estrone,[40] with various other cytochrome P450 (CYP) enzymes and metabolic transformations also being involved.[41]
Estradiol is also esterified into lipoidal estradiol forms like estradiol palmitate and estradiol stearate, which are stored in adipose tissue and may act as a very long-lasting reservoir of estradiol.[42]
2-Hydroxylation
Addition of a hydroxyl group at C2 represents the major hepatic pathway for estradiol metabolism, as mediated by CYP1A2, CYP2C8, CYP2C9, and CYP3A4. Extrahepatic 2-hydroxylation is chiefly mediated by CYP1A1 and CYP3A4.
2-Hydroxyestradiol (2-OHE2) can experience three metabolic fates: methylation to yield 2-meOHE2, oxidation to form quinones, or dehydrogenation to yield 2-OHE1.
2-OHE2 can bind to estrogen receptors but with markedly lower affinity. This metabolite has several physiological consequences: the ability to influence intracellular signalling, adenohypophyseal hormone secretion, radical and quinone formation, and inhibition of tumor formation. Weak carcinogenic activity has been shown, likely due to radical formation and induction of single-strand DNA breaks.[43]
Inactivation of 2-OHE2 is catalysed by catechol-O-methyltransferase (COMT),[44] with COMT exhibiting a faster rate for the methylation of 2-OHE2 versus 4-OH-E2. COMT, a blood-borne enzyme, mediates the most common form of 2- or 4-hydroxyestradiol inactivation, in addition to glucuronidation and sulfation. However, this inactivation can allow for the accumulation of 4-OHE2, as 2-OHE2 inhibits 4-OHE2 methylation by COMT, but 4-OHE2 does not inhibit 2-OH-E2 methylation in return.
Antitumor activity of 2-meOE2 is thought to be mediated by antiproliferative and antimetastatic effects. Inhibition of cellular proliferation and metastasis appears to be via induction of caspase-8, followed by caspase-3 and eventually DNA fragmentation. Induction of apoptosis by 2-meOE2 may be p53 dependent or independent. 2-meOE2 has also been found to inhibit aromatase activity, thereby lowering the in situ synthesis of E2 in cancer tissue.[45] 2-meOE2 has a higher binding affinity for sex hormone-binding globulin (SHBG) than E2 and 2-OH-E2 and has no affinity for the estrogen receptor.
2-meOE2 is also a potent inhibitor of angiogenesis in tumor tissues. Administration of this estradiol metabolite prevents vascular smooth muscle growth. This inhibition of angiogenesis is eliminated by co-administration with cytochrome P450 and COMT inhibitors, thereby confirming the involvement of cytochrome P450 enzymes in the blockade of tumor blood supply.
Further antitumor activity of 2-meOE2 has been identified through immunomodulation. The cytokines IL-6 and TNFα, as well the prostaglandin PGE2, are capable of stimulating aromatase activity. Since macrophages and lymphocytes are present in breast tissue, this provides a concerning means of upregulating in situ estradiol biosynthesis. 2-meOE2 appeared to be able to halve the basal aromatase activity in mammary fibroblasts, possibly through destabilisation of the microtubules that mediate translocation of the cytokine receptors to the plasma membrane. Inhibition of cytokine receptor synthesis and blockade of the autocrine and paracrine actions of cytokines and PGE2 were also observed.[46]
4-Hydroxylation

The enzyme most responsible for estradiol 4-hydroxylation is CYP1B1. In humans, CYP1B1 mRNA and protein exhibit constitutive expression in the lung and kidney, as well as estrogen-regulated tissues such as breast, ovary and uterus. Whereas 4-hydroxylation constitutes the minor pathway in the liver, the greater proportion of CYP1B1 expression in extrahepatic tissues shifts the balance in favor of 4-OH-E2 formation. 4-OH-E2 is thought to be the most carcinogenic of all the estradiol metabolites, especially considering that CYP1B1 exhibits overexpression in breast cancer tumors.
4-OH-E2, like 2-OH-E2, can be physiologically active as well as tumorigenic. 4-OH-E2 is capable of binding ER with a reduced dissociation rate and prolonged activation, thereby inducing cellular growth and proliferation,[47] adenohypophyseal hormone secretion, and prostaglandin production.
Das et al.[48] implicated 4-OH-E2 in the induction of estrogen-responsive genes, a response that exhibited partial or no abrogation by coadministration with an antiestrogen, providing evidence for the ability of 4-OH-E2 to carry out genetic upregulation via a pathway independent of ER signalling. Effects independent of ER binding include breakage of single-stranded DNA, especially when interacting synergistically with nitric oxide in human breast cancer cells and the production of quinones and free radicals.
CYP1B1 can be induced by E2.[49] ERα, after binding to estradiol, interacts with the CYP1B1 ERE to stimulate CYP1B1 expression. Thus, although E2 causes genetic changes conducive to its own inactivation, the decrease in estrogenic activity yields a toxicologically active metabolite that constitutes an additional pathway of estradiol-dependent carcinogenesis.
4-OH-E2 shares the metabolic scheme of 2-OH-E2: methylation to 4-methoxyestradiol (4-meOE2), oxidation to quinones, or dehydrogenation to 4-OH-E1. Conjugation by the ubiquitously present COMT represents the most common extrahepatic pathway of 4-OH-E2 inactivation. However, if estrogen homeostasis is imbalanced by an increase in CYP1B1 and a decrease in COMT, a greater degree of genotoxic quinone formation from 4-OH-E2 will occur.[50] 4-OHE2 can be oxidized by microsomal CYPs or peroxidases to yield estradiol-3,4-semiquinone.[51] This semiquinone can undergo redox cycling with oxygen to form estradiol-3,4-quinone (E2-3,4-Q) and superoxide. E2-3,4-Q can be converted back to 4-OHE2 in a single step by quinone reductase, or in two sequential steps catalysed by P450 reductase via the semiquinone intermediate. GSH / S-transferase activity can abrogate E2-3,4-Q levels via formation of glutathione conjugates.
E2-3,4-Q is a potent nucleophile, and will readily react with electrophilic DNA. This yields the formation of the DNA adducts 4-OHE2-1-N7Gua and 4-OHE2-1-N3Ade via a Michael addition. Destabilization of the glycosyl bond between the nitrogenous base and ribose sugar creates apurinic sites as the unstable adducts are lost from DNA. 4-OHE2-1-N7Gua has a relatively slow depurination half-life of approx. 3 hours, allowing enough time for base excision repair mechanisms to correct the change. However, 4-OHE2-1-N3Ade exhibits instantaneous depurination, leading to error-prone repair and the induction of mutations. Indeed, E2-3,4-Q has been shown to cause A-to-G mutations in the gene coding for H¬-ras, ras being vital to the correct regulation of the cellular response to growth factors. Though 2- and 4-OHE2 have similar redox potentials and thus similar redox cycling activity, the greater carcinogenic capacity of 4-OHE2 can be attributed to its increased reactivity with DNA. Another harmful effect of estrogen redox cycling is the production of superoxide and hydroxyl radicals. P450 reductase catalysis produces superoxide radicals, which can, in the presence of superoxide dismutase and Fe3+, form highly reactive hydroxyl radicals capable of damaging virtually all macromolecules.
16α-Hydroxylation
Through the action of CYP1A1, CYP1A2, CYP2C8, and the CYP3A isoforms, 16α-hydroxyestradiol (16α-OHE2), also known as estriol, is produced in abundance during pregnancy. 16α-OHE2 can be dehydrogenated to 16α-hydroxyestrone (16α-OHE1), a metabolite that has been shown to bind covalently to the estrogen receptor via Schiff base formation.[52] This covalent linkage occurs between the steroid carbonyl and the ε-amino group of lysine. In theory, 16α-OHE1 could also bind DNA, although this has not been observed. 16α-OHE2 is a potent ER agonist, capable of levels of cellular proliferation stimulation that near those obtained with E2.[53] Though studies in hamster kidney tumor models showed weak carcinogenicity, the carcinogenic potential of 16α-OHE2 in humans remains unknown.
Other hydroxylations
The function of the remainder of the hydroxylated E2 metabolites (6α-, 6β-, 7α-, 12β-, 15α-, 15β-, and 16β-OHE2) remain to be elucidated. Some of these metabolites, such as 15α-OHE2, are excreted in relatively large amounts in pregnant women, possibly serving as an indicator of good fetal health.
Levels
During the reproductive years of the human female, its serum levels are somewhat higher than that of estrone, except during the early follicular phase of the menstrual cycle; thus, estradiol may be considered the predominant estrogen during human female reproductive years in terms of absolute serum levels and estrogenic activity. During pregnancy, estriol becomes the predominant circulating estrogen, and this is the only time at which estetrol occurs in the body, while during menopause, estrone predominates (both based on serum levels). The estradiol produced by male humans, from testosterone, is present at serum levels roughly comparable to those of postmenopausal women (14-55 versus <35 pg/mL, respectively). It has also been reported that if concentrations of estradiol in a 70-year-old man are compared to those of a 70-year-old woman, levels are approximately 2- to 4-fold higher in the man.[54]
Measurement
In women, serum estradiol is measured in a clinical laboratory and reflects primarily the activity of the ovaries. As such, they are useful in the detection of baseline estrogen in women with amenorrhea or menstrual dysfunction, and to detect the state of hypoestrogenicity and menopause. Furthermore, estrogen monitoring during fertility therapy assesses follicular growth and is useful in monitoring the treatment. Estrogen-producing tumors will demonstrate persistent high levels of estradiol and other estrogens. In precocious puberty, estradiol levels are inappropriately increased.
Ranges
Individual laboratory results should always been interpreted using the ranges provided by the laboratory that performed the test.

- The ranges denoted By biological stage may be used in closely monitored menstrual cycles in regard to other markers of its biological progression, with the time scale being compressed or stretched to how much faster or slower, respectively, the cycle progresses compared to an average cycle.
- The ranges denoted Inter-cycle variability are more appropriate to use in unmonitored cycles with only the beginning of menstruation known, but where the woman accurately knows her average cycle lengths and time of ovulation, and that they are somewhat averagely regular, with the time scale being compressed or stretched to how much a woman's average cycle length is shorter or longer, respectively, than the average of the population.
- The ranges denoted Inter-woman variability are more appropriate to use when the average cycle lengths and time of ovulation are unknown, but only the beginning of menstruation is given.[55]
| Reference ranges for serum estradiol | |||
|---|---|---|---|
| Patient type | Lower limit | Upper limit | Unit |
| Adult male | 50[56] | 200[56] | pmol/L |
| 14 | 55 | pg/mL | |
| Adult female (follicular phase, day 5) | 70[56] 95% PI (standard) | 500[56] 95% PI | pmol/L |
| 110[57] 90% PI (used in diagram) | 220[57] 90% PI | ||
| 19 (95% PI) | 140 (95% PI) | pg/mL | |
| 30 (90% PI) | 60 (90% PI) | ||
| Adult female (preovulatory peak) | 400[56] | 1500[56] | pmol/L |
| 110 | 410 | pg/mL | |
| Adult female (luteal phase) | 70[56] | 600[56] | pmol/L |
| 19 | 160 | pg/mL | |
| Adult female - free (not protein bound) | 0.5[58] | 9[58] | pg/mL |
| 1.7[58] | 33[58] | pmol/L | |
| Post-menopausal female | N/A[56] | < 130[56] | pmol/L |
| N/A | < 35 | pg/mL | |
In the normal menstrual cycle, estradiol levels measure typically <50 pg/ml at menstruation, rise with follicular development (peak: 200 pg/ml), drop briefly at ovulation, and rise again during the luteal phase for a second peak. At the end of the luteal phase, estradiol levels drop to their menstrual levels unless there is a pregnancy.
During pregnancy, estrogen levels, including estradiol, rise steadily toward term. The source of these estrogens is the placenta, which aromatizes prohormones produced in the fetal adrenal gland.
Medical uses
Estradiol is used as a medication, mainly in hormone replacement therapy.[59]
Chemistry
Estradiol is an estrane (C18) steroid.[59] It is also known as 17β-estradiol (to distinguish it from 17α-estradiol) or as estra-1,3,5(10)-triene-3,17β-diol. It has two hydroxyl groups, one at the C3 position and the other at the 17β position, as well as three double bonds in the A ring. Due to its two hydroxyl groups, estradiol is often abbreviated as E2. The structurally related estrogens, estrone (E1), estriol (E3), and estetrol (E4) have one, three, and four hydroxyl groups, respectively.
History
Estradiol was first isolated in 1935.[60] It was also originally known as dihydroxyestrin or alpha-estradiol.[61][62]
Society and culture
Etymology
The name estradiol derives from estra-, Gk. οἶστρος (oistros, literally meaning "verve or inspiration"),[63] which refers to the estrane steroid ring system, and -diol, a chemical term and suffix indicating that the compound is a type of alcohol bearing two hydroxyl groups.
References
- ↑ Susan M. Ford; Sally S. Roach (7 October 2013). Roach's Introductory Clinical Pharmacology. Lippincott Williams & Wilkins. pp. 525–. ISBN 978-1-4698-3214-2.
- ↑ Maryanne Hochadel; Mosby (1 April 2015). Mosby's Drug Reference for Health Professions. Elsevier Health Sciences. pp. 602–. ISBN 978-0-323-31103-8.
- 1 2 3 4 5 Stanczyk, Frank Z.; Archer, David F.; Bhavnani, Bhagu R. (2013). "Ethinyl estradiol and 17β-estradiol in combined oral contraceptives: pharmacokinetics, pharmacodynamics and risk assessment". Contraception. 87 (6): 706–727. doi:10.1016/j.contraception.2012.12.011. ISSN 0010-7824.
- ↑ Tommaso Falcone; William W. Hurd (2007). Clinical Reproductive Medicine and Surgery. Elsevier Health Sciences. pp. 22–. ISBN 0-323-03309-1.
- ↑ Price, T; Blauer, K; Hansen, M; Stanczyk, F; Lobo, R; Bates, G (1997). "Single-dose pharmacokinetics of sublingual versus oral administration of micronized 17-estradiol". Obstetrics & Gynecology. 89 (3): 340–345. doi:10.1016/S0029-7844(96)00513-3. ISSN 0029-7844.
- ↑ Naunton, Mark; Al Hadithy, Asmar F. Y.; Brouwers, Jacobus R. B. J.; Archer, David F. (2006). "Estradiol gel". Menopause. 13 (3): 517–527. doi:10.1097/01.gme.0000191881.52175.8c. ISSN 1072-3714.
- ↑ Ryan KJ (August 1982). "Biochemistry of aromatase: significance to female reproductive physiology". Cancer Res. 42 (8 Suppl): 3342s–3344s. PMID 7083198.
- ↑ Mechoulam R, Brueggemeier RW, Denlinger DL (September 1984). "Estrogens in insects" (PDF). Cellular and Molecular Life Sciences. 40 (9): 942–944. doi:10.1007/BF01946450.
- ↑ Ozon R (1972). "Estrogens in Fishes, Amphibians, Reptiles, and Birds". In Idler DR. Steroids In Nonmammalian Vertebrates. Oxford: Elsevier Science. pp. 390–414. ISBN 032314098X.
- ↑ Saldanha, Colin J., Luke Remage-Healey, and Barney A. Schlinger. "Synaptocrine signaling: steroid synthesis and action at the synapse." Endocrine reviews 32.4 (2011): 532-549.
- ↑ Carreau S, Lambard S, Delalande C, Denis-Galeraud I, Bilinska B, Bourguiba S (2003). "Aromatase expression and role of estrogens in male gonad : a review". Reproductive Biology and Endocrinology. 1: 35. doi:10.1186/1477-7827-1-35. PMC 155680
 . PMID 12747806.
. PMID 12747806. - ↑ Pentikäinen V, Erkkilä K, Suomalainen L, Parvinen M, Dunkel L (2000). "Estradiol acts as a germ cell survival factor in the human testis in vitro". The Journal of Clinical Endocrinology and Metabolism. 85 (5): 2057–67. doi:10.1210/jcem.85.5.6600. PMID 10843196.
- ↑ Sharpe RM, Skakkebaek NE (1993). "Are oestrogens involved in falling sperm counts and disorders of the male reproductive tract?". Lancet. 341 (8857): 1392–5. doi:10.1016/0140-6736(93)90953-E. PMID 8098802.
- ↑ Handelsman, DJ (2001). "Estrogens and falling sperm counts.". Reproduction, Fertility and Development. 13 (4): 317–24. PMID 11800170.
- ↑ Fisch, Harry; Goldstein, Robert (2003). "Environmental estrogens and sperm counts" (PDF). Pure Applied Chemistry. 75 (11–12): 2181–2193. doi:10.1351/pac200375112181.
- ↑ Raman JD, Schlegel PN (2002). "Aromatase inhibitors for male infertility". The Journal of Urology. 167 (2 Pt 1): 624–9. doi:10.1016/S0022-5347(01)69099-2. PMID 11792932.
- ↑ Visootsak J, Graham JM (2006). "Klinefelter syndrome and other sex chromosomal anueploidies". Orphanet Journal of Rare Diseases. 1 (42): 42. doi:10.1186/1750-1172-1-42. PMC 1634840
 . PMID 17062147. Retrieved 20 November 2013.
. PMID 17062147. Retrieved 20 November 2013. - ↑ Carani C, Qin K, Simoni M, Faustini-Fustini M, Serpente S, Boyd J, Korach KS, Simpson ER (1997). "Effect of testosterone and estradiol in a man with aromatase deficiency". The New England Journal of Medicine. 337 (2): 91–5. doi:10.1056/NEJM199707103370204. PMID 9211678.
- ↑ Albright, Fuller; Smith Patricia H.; Richardson Anna M. (31 May 1941). "Postmenopausal Osteoporosis: Its Clinical Features". JAMA. 116 (22): 2465–2474. doi:10.1001/jama.1941.02820220007002. Retrieved 20 November 2013.
- ↑ Behl C, Widmann M, Trapp T, Holsboer F (November 1995). "17-beta estradiol protects neurons from oxidative stress-induced cell death in vitro". Biochem. Biophys. Res. Commun. 216 (2): 473–82. doi:10.1006/bbrc.1995.2647. PMID 7488136.
- ↑ Meethal, S. V.; Liu, T.; Chan, H. W.; Ginsburg, E.; Wilson, A. C.; Gray, D. N.; Bowen, R. L.; Vonderhaar, B. K.; Atwood, C. S. (2009). "Identification of a regulatory loop for the synthesis of neurosteroids: A steroidogenic acute regulatory protein-dependent mechanism involving hypothalamic-pituitary-gonadal axis receptors". Journal of Neurochemistry. 110 (3): 1014–1027. doi:10.1111/j.1471-4159.2009.06192.x. PMC 2789665
 . PMID 19493163.
. PMID 19493163. - ↑ Douma SL, Husband C, O'Donnell ME, Barwin BN, Woodend AK (2005). "Estrogen-related mood disorders: reproductive life cycle factors". Adv Nurs Sci. 28 (4): 364–75. doi:10.1097/00012272-200510000-00008. PMID 16292022.
- ↑ Lasiuk GC, Hegadoren KM (October 2007). "The effects of estradiol on central serotonergic systems and its relationship to mood in women". Biol Res Nurs. 9 (2): 147–60. doi:10.1177/1099800407305600. PMID 17909167.
- ↑ Hulshoff HE, Cohen-Kettenis PT, Van Haren NE, Peper JS, Brans RG, Cahn W, Schnack HG, Gooren LJ, Kahn RS (July 2006). "Changing your sex changes your brain: influences of testosterone and estrogen on adult human brain structure". European Journal of Endocrinology. 155 (suppl_1): 107–114. doi:10.1530/eje.1.02248.
- ↑ Harding CF (June 2004). "Hormonal Modulation of Singing: Hormonal Modulation of the Songbird Brain and Singing Behavior". Ann. N.Y. Acad. Sci. The New York Academy of Sciences. 1016: 524–539. doi:10.1196/annals.1298.030. PMID 15313793. Retrieved 2007-03-07.
- ↑ Simerly RB (2002-03-27). "Wired for reproduction: organization and development of sexually dimorphic circuits in the mammalian forebrain" (pdf). Annu. Rev. Neurosci. 25: 507–536. doi:10.1146/annurev.neuro.25.112701.142745. PMID 12052919. Retrieved 2007-03-07.
- ↑ Bulzomi P, Bolli A, Galluzzo P, Leone S, Acconcia F, Marino M (January 2010). "Naringenin and 17β-estradiol coadministration prevents hormone-induced human cancer cell growth". IUBMD Life. 62 (1): 51–60. doi:10.1002/iub.279. PMID 19960539.
- ↑ Sreeja S, Santhosh Kumar TR, Lakshmi BS, Sreeja S (17 March 2011). "Pomegranate extract demonstrate a selective estrogen receptor modulator profile in human tumor cell lines and in vivo models of estrogen deprivation". Journal of Nutritional Biochemistry. 23 (7): 725–32. doi:10.1016/j.jnutbio.2011.03.015. PMID 21839626.
- ↑ Thomas CG, Strom A, Lindberg K, Gustafsson JA (22 June 2010). "Estrogen receptor beta decreases survival of p53-defective cancer cells after DNA damage by impairing G2/M checkpoint signaling". Breast Cancer Research and Treatment. 127 (2): 417–427. doi:10.1007/s10549-010-1011-z. PMID 20623183.
- ↑ Collins P, Rosano GM, Sarrel PM, Ulrich L, Adamopoulos S, Beale CM, McNeill JG, Poole-Wilson PA (1995). "17 beta-Estradiol attenuates acetylcholine-induced coronary arterial constriction in women but not men with coronary heart disease". Circulation. 92 (1): 24–30. doi:10.1161/01.CIR.92.1.24. PMID 7788912.
- ↑ Prossnitz ER, Barton M (May 2014). "Estrogen biology: New insights into GPER function and clinical opportunities". Mol. Cell. Endocrinol. 389 (1–2): 71–83. doi:10.1016/j.mce.2014.02.002. PMC 4040308
 . PMID 24530924.
. PMID 24530924. - ↑ Prossnitz ER, Arterburn JB, Sklar LA (2007). "GPR30: A G protein-coupled receptor for estrogen". Mol. Cell. Endocrinol. 265-266: 138–42. doi:10.1016/j.mce.2006.12.010. PMC 1847610
 . PMID 17222505.
. PMID 17222505. - ↑ Soltysik K, Czekaj P (April 2013). "Membrane estrogen receptors - is it an alternative way of estrogen action?". J. Physiol. Pharmacol. 64 (2): 129–42. PMID 23756388.
- ↑ Micevych PE, Kelly MJ (2012). "Membrane estrogen receptor regulation of hypothalamic function". Neuroendocrinology. 96 (2): 103–10. doi:10.1159/000338400. PMC 3496782
 . PMID 22538318.
. PMID 22538318. - ↑ Häggström, Mikael; Richfield, David (2014). "Diagram of the pathways of human steroidogenesis". WikiJournal of Medicine. 1 (1). doi:10.15347/wjm/2014.005. ISSN 2002-4436.
- ↑ Walter F. Boron; Emile L. Boulpaep (2003). Medical Physiology: A Cellular And Molecular Approach. Elsevier/Saunders. p. 1300. ISBN 1-4160-2328-3.
- ↑ Mutschler, Ernst; Schäfer-Korting, Monika (2001). Arzneimittelwirkungen (in German) (8 ed.). Stuttgart: Wissenschaftliche Verlagsgesellschaft. pp. 434, 444. ISBN 3-8047-1763-2.
- ↑ Young, I. J.; Hillman, J. R.; Knights, B. A. (1978). "Endogenous Estradiol-17 β in Phaseolus vulgaris". Zeitschrift für Pflanzenphysiologie. 90: 45–50. doi:10.1016/S0044-328X(78)80223-2.
- ↑ Wu CH, Motohashi T, Abdel-Rahman HA, Flickinger GL, Mikhail G (August 1976). "Free and protein-bound plasma estradiol-17 beta during the menstrual cycle". J. Clin. Endocrinol. Metab. 43 (2): 436–45. doi:10.1210/jcem-43-2-436. PMID 950372.
- ↑ Cheng ZN, Shu Y, Liu ZQ, Wang LS, Ou-Yang DS, Zhou HH (February 2001). "Role of cytochrome P450 in estradiol metabolism in vitro". Acta Pharmacol. Sin. 22 (2): 148–54. PMID 11741520.
- ↑ Lee AJ, Cai MX, Thomas PE, Conney AH, Zhu BT (August 2003). "Characterization of the oxidative metabolites of 17beta-estradiol and estrone formed by 15 selectively expressed human cytochrome p450 isoforms". Endocrinology. 144 (8): 3382–98. doi:10.1210/en.2003-0192. PMID 12865317.
- ↑ Michael Oettel; Ekkehard Schillinger (6 December 2012). Estrogens and Antiestrogens II: Pharmacology and Clinical Application of Estrogens and Antiestrogen. Springer Science & Business Media. pp. 235–237,268,271. ISBN 978-3-642-60107-1.
- ↑ Liehr, J. G. (1 February 2000). "Is Estradiol a Genotoxic Mutagenic Carcinogen?". Endocrine Reviews. 21 (1): 40–54. doi:10.1210/EDRV.21.1.0386. PMID 10696569.
- ↑ Li, K.-M. (24 October 2003). "Metabolism and DNA binding studies of 4-hydroxyestradiol and estradiol-3,4-quinone in vitro and in female ACI rat mammary gland in vivo". Carcinogenesis. 25 (2): 289–297. doi:10.1093/carcin/bgg191. PMID 14578156.
- ↑ Lakhani, NJ; Sarkar, MA; Venitz, J; Figg, WD (February 2003). "2-Methoxyestradiol, a promising anticancer agent.". Pharmacotherapy. 23 (2): 165–172. doi:10.1592/phco.23.2.165.32088. PMID 12587805.
- ↑ Purohit, A.; Singh, A.; Ghilchik, M.W.; Reed, M.J. (July 1999). "Inhibition of Tumor Necrosis Factor α-Stimulated Aromatase Activity by Microtubule-Stabilizing Agents, Paclitaxel and 2-Methoxyestradiol". Biochemical and Biophysical Research Communications. 261 (1): 214–217. doi:10.1006/bbrc.1999.1010. PMID 10405348.
- ↑ Cheng, Z. N.; Shu, Y.; Liu, Z. Q.; Wang, L. S.; Ou-Yang, D. S.; Zhou, H. H. (2001). "Role of cytochrome P450 in estradiol metabolism in vitro" (PDF). Acta pharmacologica Sinica. 22 (2): 148–154. PMID 11741520.
- ↑ Das, SK; Taylor, JA; Korach, KS; Paria, BC; Dey, SK; Lubahn, DB (25 November 1997). "Estrogenic responses in estrogen receptor-alpha deficient mice reveal a distinct estrogen signaling pathway.". Proceedings of the National Academy of Sciences of the United States of America. 94 (24): 12786–91. doi:10.1073/pnas.94.24.12786. PMC 24216
 . PMID 9371753.
. PMID 9371753. - ↑ Tsuchiya, Y. (1 May 2004). "Human CYP1B1 Is Regulated by Estradiol via Estrogen Receptor". Cancer Research. 64 (9): 3119–3125. doi:10.1158/0008-5472.CAN-04-0166. PMID 15126349.
- ↑ Lu, Fang; Zahid, Muhammad; Saeed, Muhammad; Cavalieri, Ercole L.; Rogan, Eleanor G. (June 2007). "Estrogen metabolism and formation of estrogen-DNA adducts in estradiol-treated MCF-10F cells". The Journal of Steroid Biochemistry and Molecular Biology. 105 (1–5): 150–158. doi:10.1016/j.jsbmb.2006.12.102. PMC 1986824
 . PMID 17582757.
. PMID 17582757. - ↑ Liehr, JG; Ulubelen, AA; Strobel, HW (25 December 1986). "Cytochrome P-450-mediated redox cycling of estrogens.". The Journal of Biological Chemistry. 261 (36): 16865–70. PMID 3782146.
- ↑ Swaneck, GE; Fishman, J (November 1988). "Covalent binding of the endogenous estrogen 16 alpha-hydroxyestrone to estradiol receptor in human breast cancer cells: characterization and intranuclear localization.". Proceedings of the National Academy of Sciences of the United States of America. 85 (21): 7831–5. doi:10.1073/pnas.85.21.7831. PMC 282290
 . PMID 3186693.
. PMID 3186693. - ↑ Gupta, Mona; McDougal, Andrew; Safe, Stephen (December 1998). "Estrogenic and antiestrogenic activities of 16α- and 2-hydroxy metabolites of 17β-estradiol in MCF-7 and T47D human breast cancer cells". The Journal of Steroid Biochemistry and Molecular Biology. 67 (5–6): 413–419. doi:10.1016/S0960-0760(98)00135-6. PMID 10030690.
- ↑ Sayed Y, Taxel P (2003). "The use of estrogen therapy in men". Curr Opin Pharmacol. 3 (6): 650–4. PMID 14644018.
- ↑ Häggström, Mikael (2014). "Reference ranges for estradiol, progesterone, luteinizing hormone and follicle-stimulating hormone during the menstrual cycle". WikiJournal of Medicine. 1 (1). doi:10.15347/wjm/2014.001. ISSN 2002-4436.
- 1 2 3 4 5 6 7 8 9 10 GPNotebook — reference range (oestradiol) Retrieved on September 27, 2009
- 1 2 Values taken from day 1 after LH surge in: Stricker R, Eberhart R, Chevailler MC, Quinn FA, Bischof P, Stricker R (2006). "Establishment of detailed reference values for luteinizing hormone, follicle stimulating hormone, estradiol, and progesterone during different phases of the menstrual cycle on the Abbott ARCHITECT analyzer". Clin. Chem. Lab. Med. 44 (7): 883–7. doi:10.1515/CCLM.2006.160. PMID 16776638. as PDF
- 1 2 3 4 Total amount multiplied by 0.022 according to 2.2% presented in: Wu CH, Motohashi T, Abdel-Rahman HA, Flickinger GL, Mikhail G (August 1976). "Free and protein-bound plasma estradiol-17 beta during the menstrual cycle". J. Clin. Endocrinol. Metab. 43 (2): 436–45. doi:10.1210/jcem-43-2-436. PMID 950372.
- 1 2 Kuhl H (2005). "Pharmacology of estrogens and progestogens: influence of different routes of administration". Climacteric. 8 Suppl 1: 3–63. doi:10.1080/13697130500148875. PMID 16112947.
- ↑ Fritz F. Parl (2000). Estrogens, Estrogen Receptor and Breast Cancer. IOS Press. pp. 4–. ISBN 978-0-9673355-4-4.
- ↑ Fluhmann CF (1938). "Estrogenic Hormones: Their Clinical Usage". Cal West Med. 49 (5): 362–6. PMC 1659459
 . PMID 18744783.
. PMID 18744783. - ↑ Reilly WA (1941). "Estrogens: Their Use in Pediatrics". Cal West Med. 55 (5): 237–9. PMC 1634235
 . PMID 18746057.
. PMID 18746057. - ↑ "Greek Word Study Tool: oistros". Perseus Digital Library. Retrieved 2011-12-28.
External links
- Estradiol MS Spectrum
- Estrogens - Lab Tests Online
